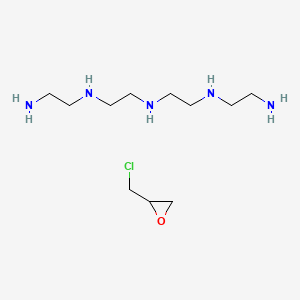
Details of Drug
| Drug-Drug Interaction Network | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sunburst Graph |
AM001 Affected gastrointestinal absorption
BM003 Complex formation
BM008 Altered Fat-soluble drugs absorption
|
||||||||
| Relation Graph | |||||||||
| Full list of drugs interacting with Colestipol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Affected gastrointestinal absorption | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complex formation | Drug Num: 64 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1094
|
Mycophenolate mofetil
|
C23H31NO7
|
|
Major | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1095
|
Mycophenolic acid
|
C17H20O6
|
|
Major | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1556
|
Teriflunomide
|
C12H9F3N2O2
|
|
Major | Inter Info |
[2]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0326
|
Chlorthalidone
|
C14H11ClN2O4S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0158
|
Bendroflumethiazide
|
C15H14F3N3O4S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0166
|
Benzthiazide
|
C15H14ClN3O4S3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0215
|
Budesonide
|
C25H34O6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0217
|
Bumetanide
|
C17H20N2O5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0303
|
Cerivastatin
|
C26H34FNO5
|
|
Moderate | Inter Info |
[15]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0310
|
Chenodeoxycholic acid
|
C24H40O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0321
|
Chlorothiazide
|
C7H6ClN3O4S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0330
|
Cholic Acid
|
C24H40O5
|
|
Moderate | Inter Info |
[17]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0432
|
Deferasirox
|
C21H15N3O4
|
|
Moderate | Inter Info |
[18]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0436
|
Deflazacort
|
C25H31NO6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0453
|
Dexamethasone
|
C22H29FO5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0490
|
Digitoxin
|
C41H64O13
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0491
|
Digoxin
|
C41H64O14
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0536
|
Drospirenone
|
C24H30O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0605
|
Esterified estrogens
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0606
|
Estradiol
|
C18H24O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0609
|
Estrone
|
C18H22O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0610
|
Estrone sulfate
|
C18H22O5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0613
|
Etacrynic acid
|
C13H12Cl2O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0619
|
Ethinylestradiol
|
C20H24O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0642
|
Fenofibrate
|
C20H21ClO4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0643
|
Fenofibric acid
|
C17H15ClO4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0671
|
Fludrocortisone
|
C21H29FO5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0686
|
Fluvastatin
|
C24H26FNO4
|
|
Moderate | Inter Info |
[15]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0704
|
Furosemide
|
C12H11ClN2O5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0773
|
Hydrocortisone
|
C21H30O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0775
|
Hydroflumethiazide
|
C8H8F3N3O4S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0798
|
Indapamide
|
C16H16ClN3O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0944
|
Lomitapide
|
C39H37F6N3O2
|
|
Moderate | Inter Info |
[36]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0990
|
Medroxyprogesterone acetate
|
C24H34O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1041
|
Methylprednisolone
|
C22H30O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1048
|
Metolazone
|
C16H16ClN3O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1154
|
Norethisterone
|
C20H26O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1157
|
Norgestrel
|
C21H28O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1160
|
Obeticholic acid
|
C26H44O4
|
|
Moderate | Inter Info |
[37]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1311
|
Polythiazide
|
C11H13ClF3N3O4S3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1332
|
Prednisolone
|
C21H28O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1334
|
Prednisone
|
C21H26O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1348
|
Progesterone
|
C21H30O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1370
|
Quinestrol
|
C25H32O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1376
|
Raloxifene
|
C28H27NO4S
|
|
Moderate | Inter Info |
[38]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1473
|
Simvastatin
|
C25H38O5
|
|
Moderate | Inter Info |
[15]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1628
|
Triamcinolone
|
C21H27FO6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1633
|
Trichlormethiazide
|
C8H8Cl3N3O4S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1688
|
Venetoclax
|
C45H50ClN7O7S
|
|
Moderate | Inter Info |
[2]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0387
|
Conjugated estrogens
|
C18H21NaO5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0487
|
Diethylstilbestrol
|
C18H20O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0626
|
Etonogestrel
|
C22H28O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0922
|
Levonorgestrel
|
C21H28O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0993
|
Megestrol acetate
|
C24H32O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1329
|
Pravastatin
|
C23H36O7
|
|
Moderate | Inter Info |
[15]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1611
|
Torasemide
|
C16H20N4O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0087
|
Anisindione
|
C16H12O3
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0441
|
Demeclocycline
|
C21H21ClN2O8
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1070
|
Minocycline
|
C23H27N3O7
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1209
|
Oxytetracycline
|
C22H24N2O9
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1564
|
Tetracycline
|
C22H24N2O8
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1711
|
Warfarin
|
C19H16O4
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0481
|
Dicoumarol
|
C19H12O6
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0530
|
Doxycycline
|
C22H24N2O8
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Altered Fat-soluble drugs absorption | Drug Num: 14 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0233
|
Calcifediol
|
C27H44O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0234
|
Calcitriol
|
C27H44O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0495
|
Dihydrotachysterol
|
C28H46O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0527
|
Doxercalciferol
|
C28H44O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0715
|
Gemfibrozil
|
C15H22O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0925
|
Levothyroxine
|
C15H11I4NO4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0935
|
Liotrix
|
C30H21I7N2Na2O8
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1226
|
Paricalcitol
|
C27H44O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1284
|
Phylloquinone
|
C31H46O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1577
|
Thyroid, porcine
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0591
|
Ergocalciferol
|
C28H44O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0934
|
Liothyronine
|
C15H12I3NO4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1702
|
Vitamin A
|
C20H30O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1703
|
Vitamin E
|
C29H50O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||