Details of Drug-Drug Interaction
| Drug General Information (ID: DDIYFXLNCH) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Omacetaxine mepesuccinate | Drug Info | Samarium (153Sm) lexidronam | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Therapeutic Radiopharmaceuticals | |||||||
| Structure | |||||||||
| Mechanism of Omacetaxine mepesuccinate-Samarium (153Sm) lexidronam Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
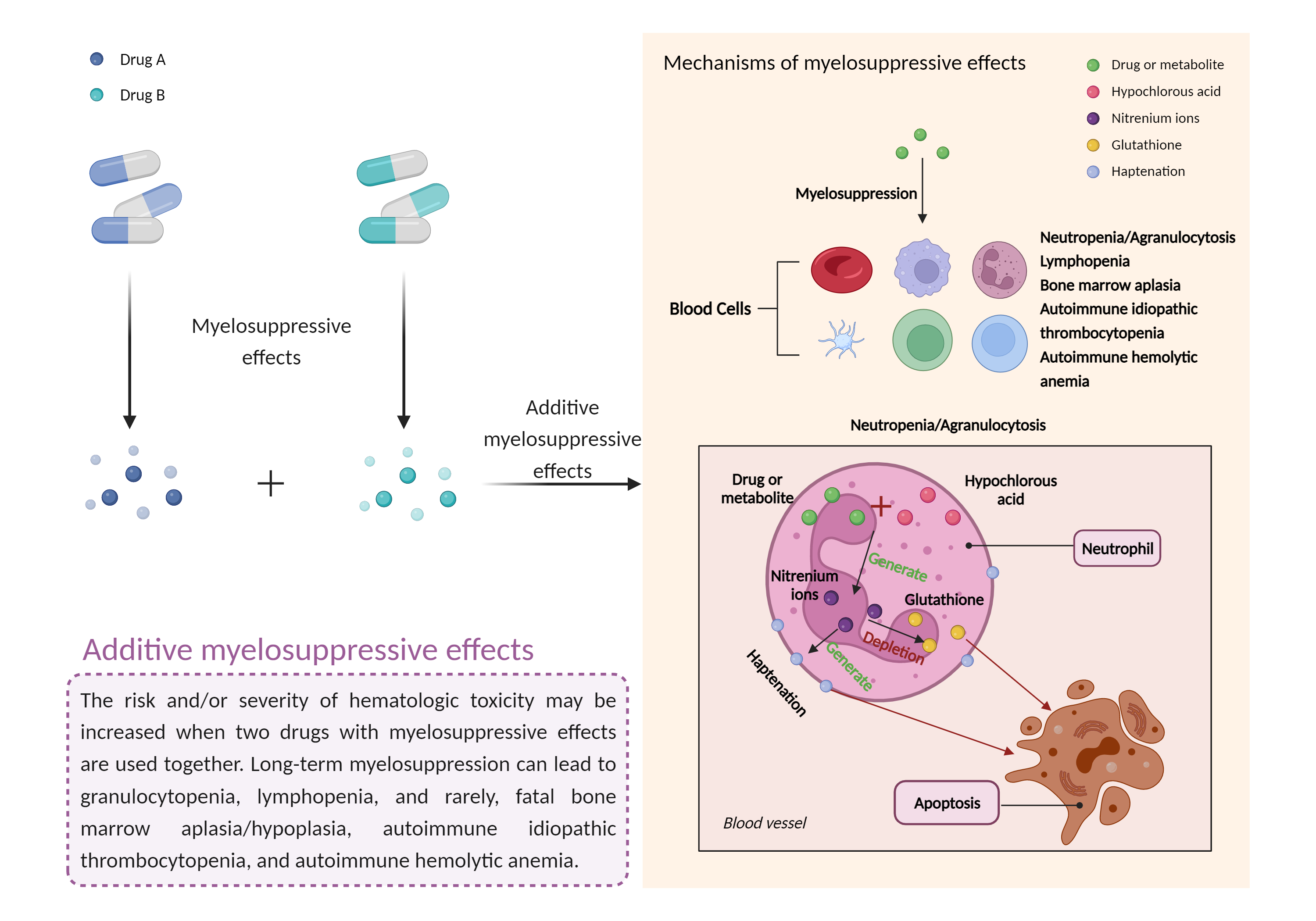
| Additive myelosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Omacetaxine mepesuccinate | Samarium (153Sm) lexidronam | |||||||
| Mechanism | Myelosuppressive effects | Myelosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Myelosuppressive effects | ||||||||
| Factor Description | Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The manufacturer recommends avoiding concomitant use of samarium sm 153 lexidronam with chemotherapy or external beam radiation therapy unless benefits are anticipated to outweigh the risks. Moreover, samarium sm 153 lexidronam should not be given after either of these treatments until there has been time for adequate marrow recovery. Caution and close monitoring of bone marrow function are advisable if coadministration with other myelotoxic agents is required. Patients should be advised to contact their physician if they develop signs and symptoms of myelosuppression such as pallor, dizziness, fatigue, lethargy, fainting, unusual bleeding or bruising, or signs of infection such as fever, chills, diarrhea, sore throat, muscle aches, shortness of breath, blood in phlegm, weight loss, red or inflamed skin, body sores, and pain or burning during urination. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Quadramet (samarium sm 153 lexidronam) Berlex Laboratories, Richmond, CA. | ||||||||||||||||||
