Details of Drug-Drug Interaction
| Drug General Information (ID: DDIXD5S7F6) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Amprenavir (oral solution) | Drug Info | Tinidazole | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anti-Hiv Agents | Amebicides/Antibiotics | |||||||
| Structure | |||||||||
| Mechanism of Amprenavir (oral solution)-Tinidazole Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
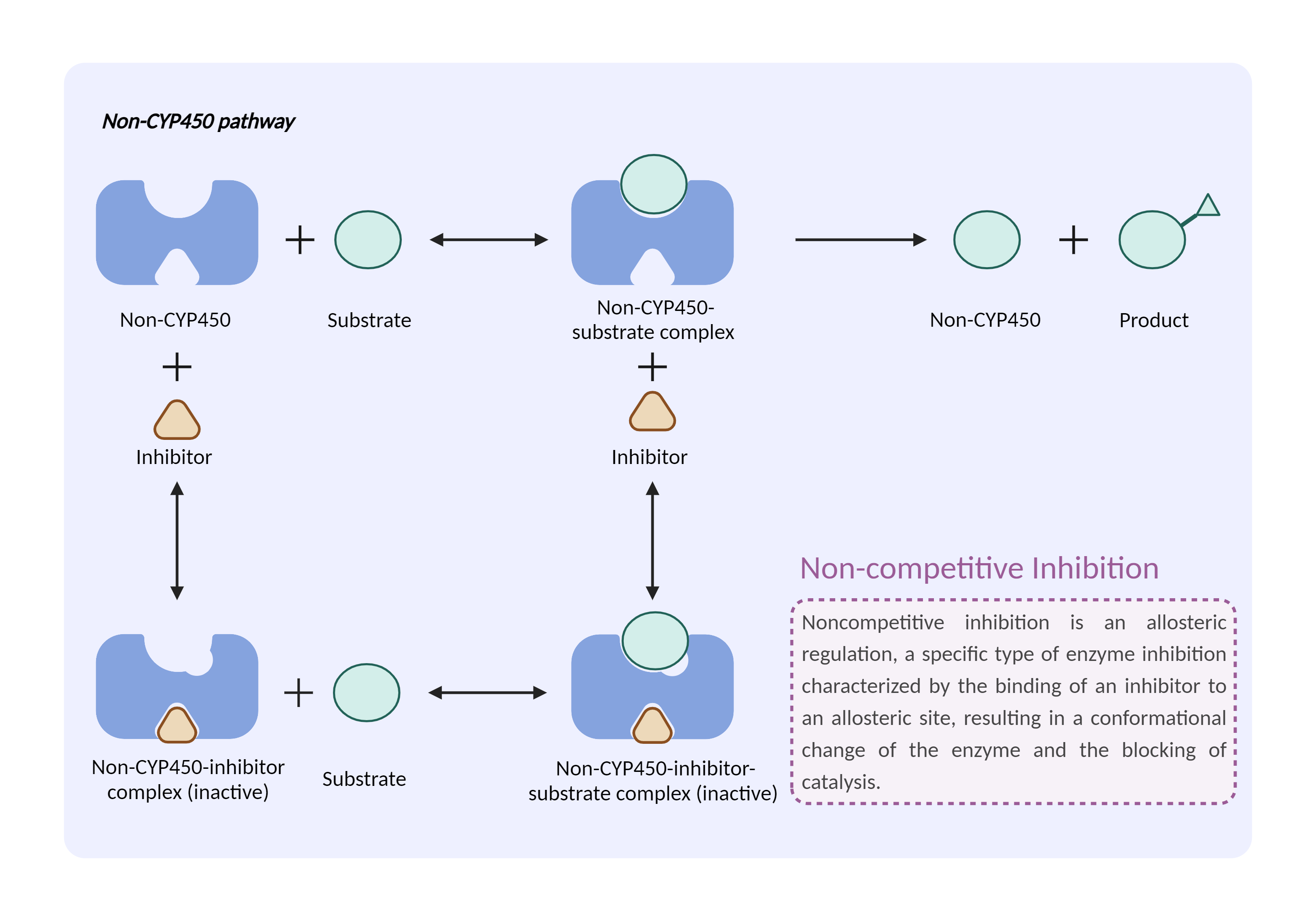
| Non-CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Amprenavir (oral solution) | Tinidazole | |||||||
| Mechanism | Aldehyde dehydrogenase substrate | Aldehyde dehydrogenase inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Aldehyde dehydrogenase 1 |
×
Structure
Sequence
MSSSGTPDLPVLLTDLKIQYTKIFINNEWHDSVSGKKFPVFNPATEEELCQVEEGDKEDVDKAVKAARQAFQIGSPWRTMDASERGRLLYKLADLIERDRLLLATMESMNGGKLYSNAYLNDLAGCIKTLRYCAGWADKIQGRTIPIDGNFFTYTRHEPIGVCGQIIPWNFPLVMLIWKIGPALSCGNTVVVKPAEQTPLTALHVASLIKEAGFPPGVVNIVPGYGPTAGAAISSHMDIDKVAFTGSTEVGKLIKEAAGKSNLKRVTLELGGKSPCIVLADADLDNAVEFAHHGVFYHQGQCCIAASRIFVEESIYDEFVRRSVERAKKYILGNPLTPGVTQGPQIDKEQYDKILDLIESGKKEGAKLECGGGPWGNKGYFVQPTVFSNVTDEMRIAKEEIFGPVQQIMKFKSLDDVIKRANNTFYGLSAGVFTKDIDKAITISSALQAGTVWVNCYGVVSAQCPFGGFKMSGNGRELGEYGFHEYTEVKTVTVKISQKNS
|
|||||||
| Gene Name | ALDH1A1 | ||||||||
| Uniprot ID | AL1A1_HUMAN | ||||||||
| KEGG Pathway | hsa:216 | ||||||||
| Protein Family | Aldehyde dehydrogenase family | ||||||||
| Protein Function |
Cytosolic dehydrogenase that catalyzes the irreversible oxidation of a wide range of aldehydes to their corresponding carboxylic acid (PubMed:19296407, PubMed:12941160, PubMed:15623782, PubMed:17175089, PubMed:26373694, PubMed:25450233). Functions downstream of retinol dehydrogenases and catalyzes the oxidation of retinaldehyde into retinoic acid, the second step in the oxidation of retinol/vitamin A into retinoic acid (By similarity). This pathway is crucial to control the levels of retinol and retinoic acid, two important molecules which excess can be teratogenic and cytotoxic (By similarity). Also oxidizes aldehydes resulting from lipid peroxidation like (E)-4-hydroxynon-2-enal/HNE, malonaldehyde and hexanal that form protein adducts and are highly cytotoxic. By participating for instance to the clearance of (E)-4-hydroxynon-2-enal/HNE in the lens epithelium prevents the formation of HNE-protein adducts and lens opacification (PubMed:19296407, PubMed:12941160, PubMed:15623782). Functions also downstream of fructosamine-3-kinase in the fructosamine degradation pathway by catalyzing the oxidation of 3-deoxyglucosone, the carbohydrate product of fructosamine 3-phosphate decomposition, which is itself a potent glycating agent that may react with lysine and arginine side-chains of proteins (PubMed:17175089). Has also an aminobutyraldehyde dehydrogenase activity and is probably part of an alternative pathway for the biosynthesis of GABA/4-aminobutanoate in midbrain, thereby playing a role in GABAergic synaptic transmission (By similarity).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The use of amprenavir oral solution with disulfiram or metronidazole (oral, intravenous, and probably vaginal preparations) is considered contraindicated. Given their structural similarities to metronidazole, the same precaution may be applicable to other nitroimidazoles such as benznidazole and tinidazole, although clinical data are lacking. | ||||||||
