Details of Drug-Drug Interaction
| Drug General Information (ID: DDIWTOKFAH) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Trandolapril | Drug Info | Iron Dextran | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Iron Supplement | |||||||
| Structure | |||||||||
| Mechanism of Trandolapril-Iron Dextran Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
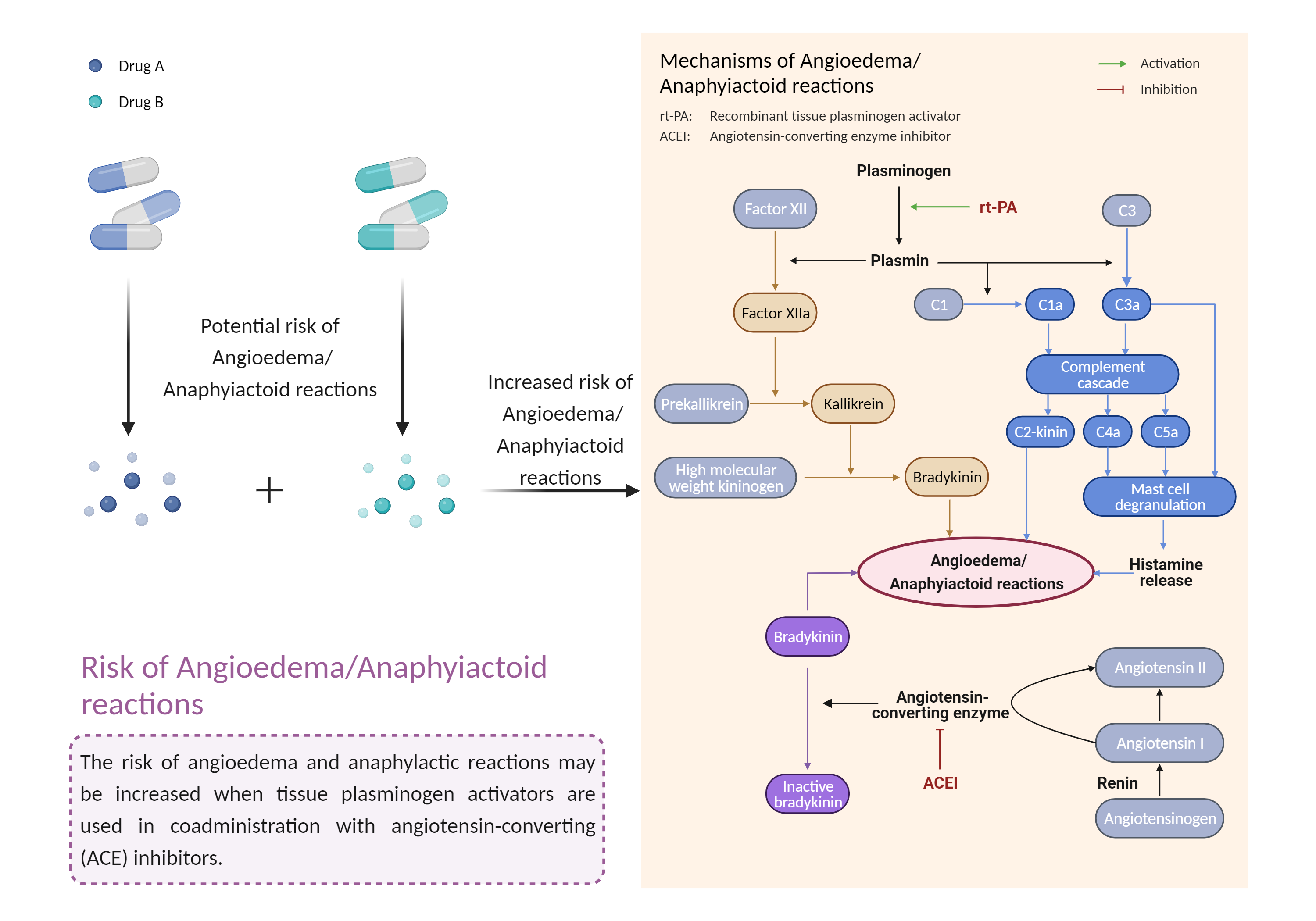
| Increased risk of angioedema/anaphylactoid reactions Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Trandolapril | Iron Dextran | |||||||
| Mechanism | Angioedema | Anaphylactoid reactions | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Angioedema/anaphylactoid reactions | ||||||||
| Factor Description | Anaphylactoid reaction is a condition in which symptoms similar to an allergic reaction occur, but no immune sensitization is detected. Most commonly, the syndrome begins with skin involvement, usually pruritus, flushing, urticaria, or angioedema. Angioedema is a reaction to a trigger that causes swelling of the tissue below the inner layer of the skin, called the dermis or submucosa, and the swelling can occur on the face, throat, or around the genitals. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Patients should be closely monitored during and after parenteral administration of iron dextran, regardless of whether they are being treated with an ACE inhibitor. This is particularly important in patients with a history of drug allergy or multiple drug allergies or an immune/inflammatory condition such as systemic lupus erythematosus or rheumatoid arthritis. Resuscitation techniques and personnel trained in the detection and treatment of anaphylactic-type reactions should be readily available. Prior to the first therapeutic dose, a test dose corresponding to 25 mg iron should be injected gradually. Although anaphylactic reactions are usually evident within a few minutes, observe patients for at least one hour before administering the remainder of the therapeutic dose. Administration must be stopped immediately if signs of an anaphylactoid reaction are observed. Patients should also be closely monitored during each subsequent administration of iron dextran. Fatal reactions have occurred following the test dose and also in situations where the test dose was tolerated. According to some studies, non-dextran parenteral iron formulations may be associated with a lower risk of adverse effects, especially death and life-threatening reactions such as anaphylaxis, cardiac arrest, and respiratory depression. | ||||||||
