Details of Drug-Drug Interaction
| Drug General Information (ID: DDIV5W6F4B) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Lidocaine | Drug Info | Tocainide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anesthetics | Antiarrhythmic Agents | |||||||
| Structure | |||||||||
| Mechanism of Lidocaine-Tocainide Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
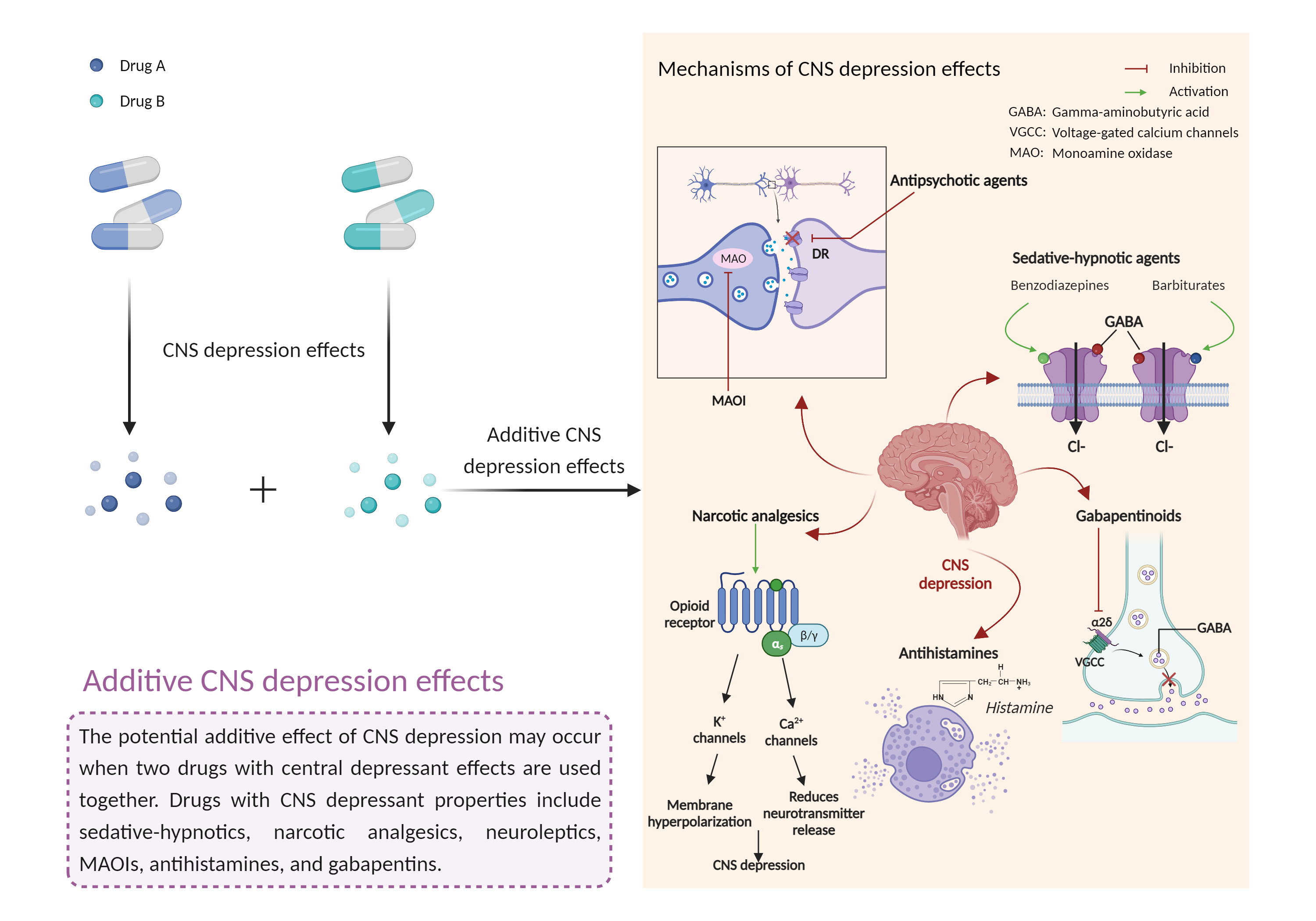
| Additive CNS depression effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Lidocaine | Tocainide | |||||||
| Mechanism | CNS depression effects | CNS depression effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | CNS depression effects | ||||||||
| Factor Description | CNS depressants are drugs that inhibit or suppress brain activity and can reduce mental and physical processes. Excessive CNS depression can lead to decreased heart rate, slow breathing (less than 10 breaths per minute), extreme confusion or loss of memory, nausea and vomiting, poor judgment, blue lips or fingertips, irritability and aggression, and clammy or cold skin. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Management consists of cautiously starting patients on tocainide six hours before stopping lidocaine infusions, then starting the next maintenance dose of tocainide six hours after the lidocaine drip has ended. The patient's heart rhythm and clinical response should be monitored during this time. | ||||||||
