Details of Drug-Drug Interaction
| Drug General Information (ID: DDITY2AJ7Z) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Didanosine | Drug Info | Elvitegravir | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anti-Hiv Agents | Antiviral Agents | |||||||
| Structure | |||||||||
| Mechanism of Didanosine-Elvitegravir Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
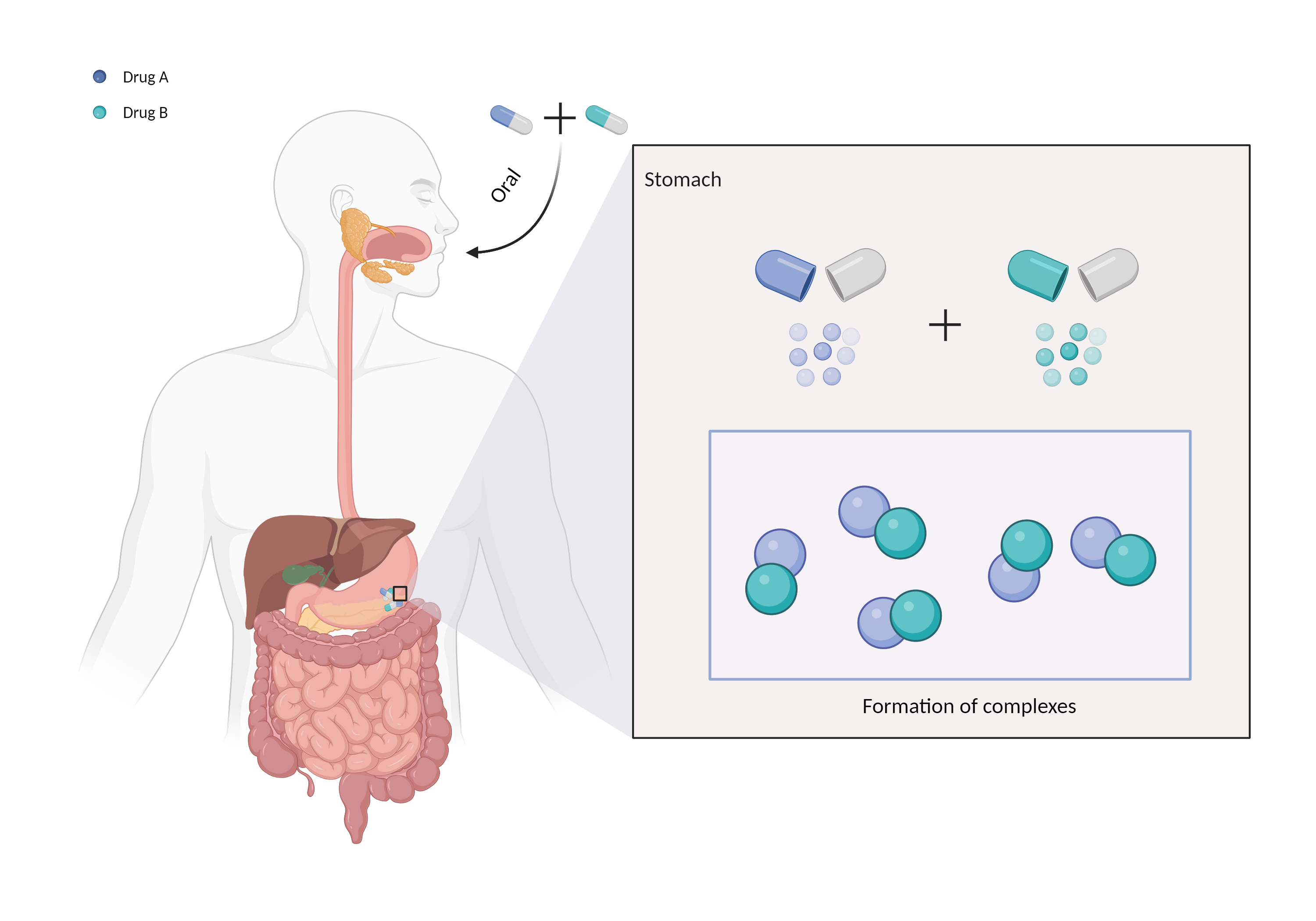
| Complex formation Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Didanosine | Elvitegravir | |||||||
| Mechanism | Polyvalent cations | Binds to polyvalent cations | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Chelation | ||||||||
| Factor Description | Chelation is a direct drug interaction that usually involves the formation of dimers or trimers, resulting in larger complexes and poorer absorption. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | When prescribed in combination with elvitegravir, didanosine should be administered on an empty stomach one hour before or two hours after elvitegravir, which is to be taken with food. Clinical monitoring for altered therapeutic efficacy is advisable. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "Australian Product Information.". | ||||||||||||||||||
| 2 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
