| Mechanism of Mazindol-Solriamfetol Interaction
(Severity Level: Moderate)
|
|
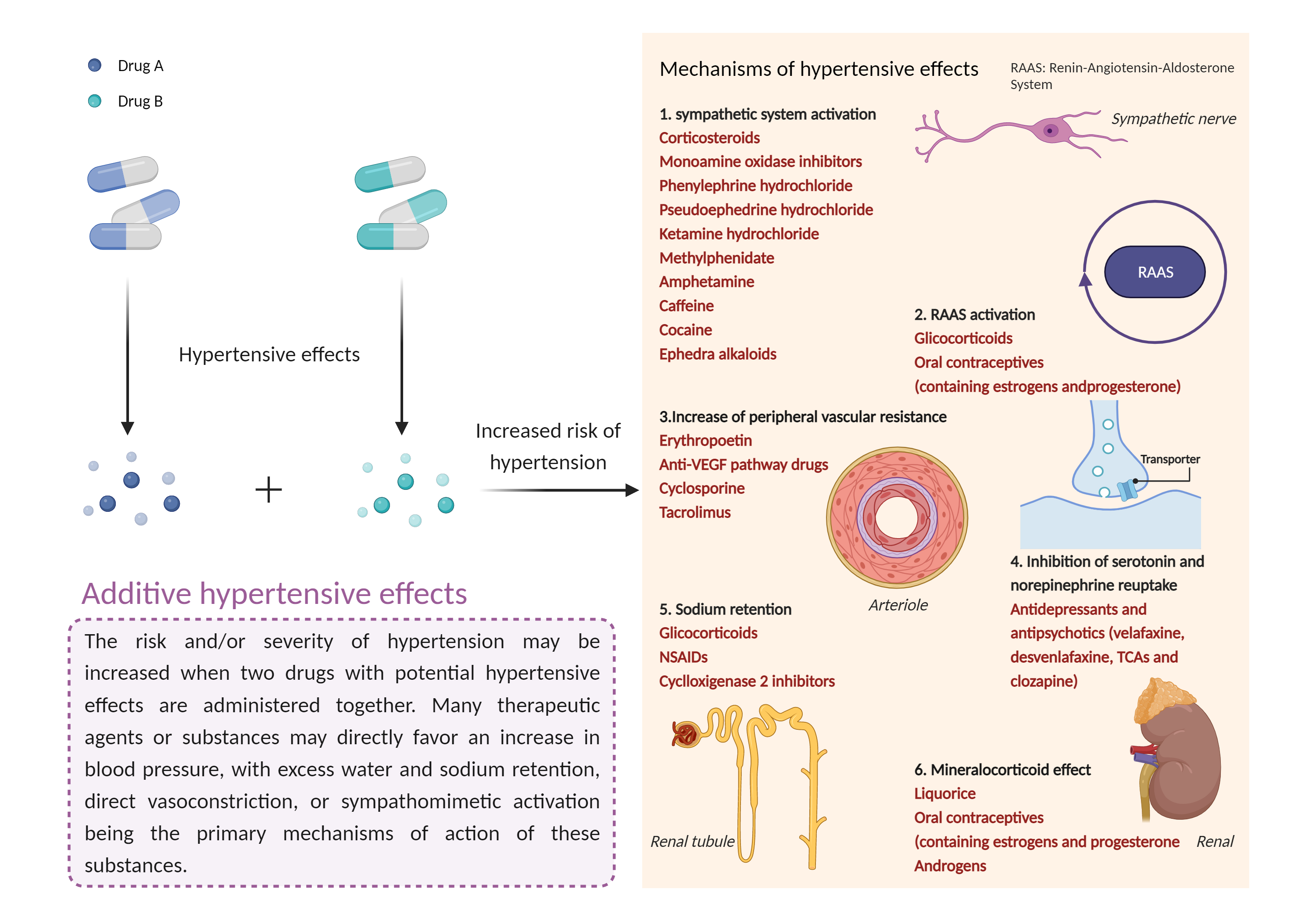
Additive hypertensive effects
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Mazindol |
Solriamfetol |
|
Mechanism 1
|
Hypertensive effects |
Hypertensive effects |
| Key Mechanism Factor 1 |
| Factor Name |
Hypertensive effects |
| Factor Description |
Hypertension is defined as a blood pressure reading above a prescribed limit (130/90 mmHg). Most people with hypertension have no signs or symptoms; others may experience: severe headache, shortness of breath, nosebleeds, severe anxiety, and feeling a tingling sensation in the neck or head. |
| Mechanism Description |
- Additive hypertensive effects by the combination of Mazindol and Solriamfetol
|
|
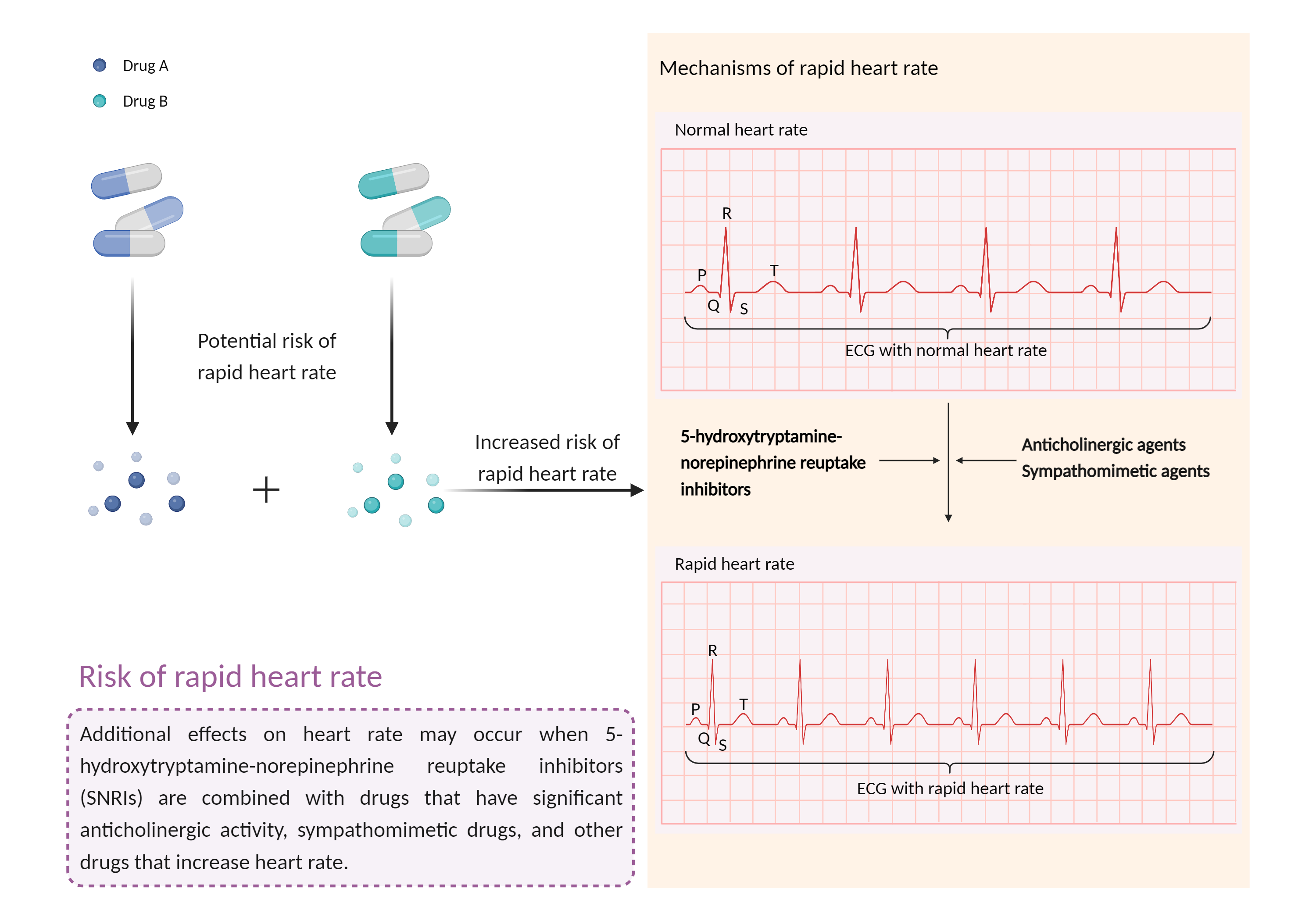
Increased risk of rapid heart rate
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Mazindol |
Solriamfetol |
|
Mechanism 2
|
Increased heart rate |
Increased heart rate |
| Key Mechanism Factor 2 |
| Factor Name |
Tachycardia |
| Factor Description |
A faster-than-normal heart rate, but the heart is beating normally. Tachycardia may not cause any symptoms or complications, but if left untreated, some forms of tachycardia can lead to serious health problems, including heart failure, stroke, or sudden cardiac death. |
| Mechanism Description |
- Increased risk of rapid heart rate by the combination of Mazindol and Solriamfetol
|


