| Management |
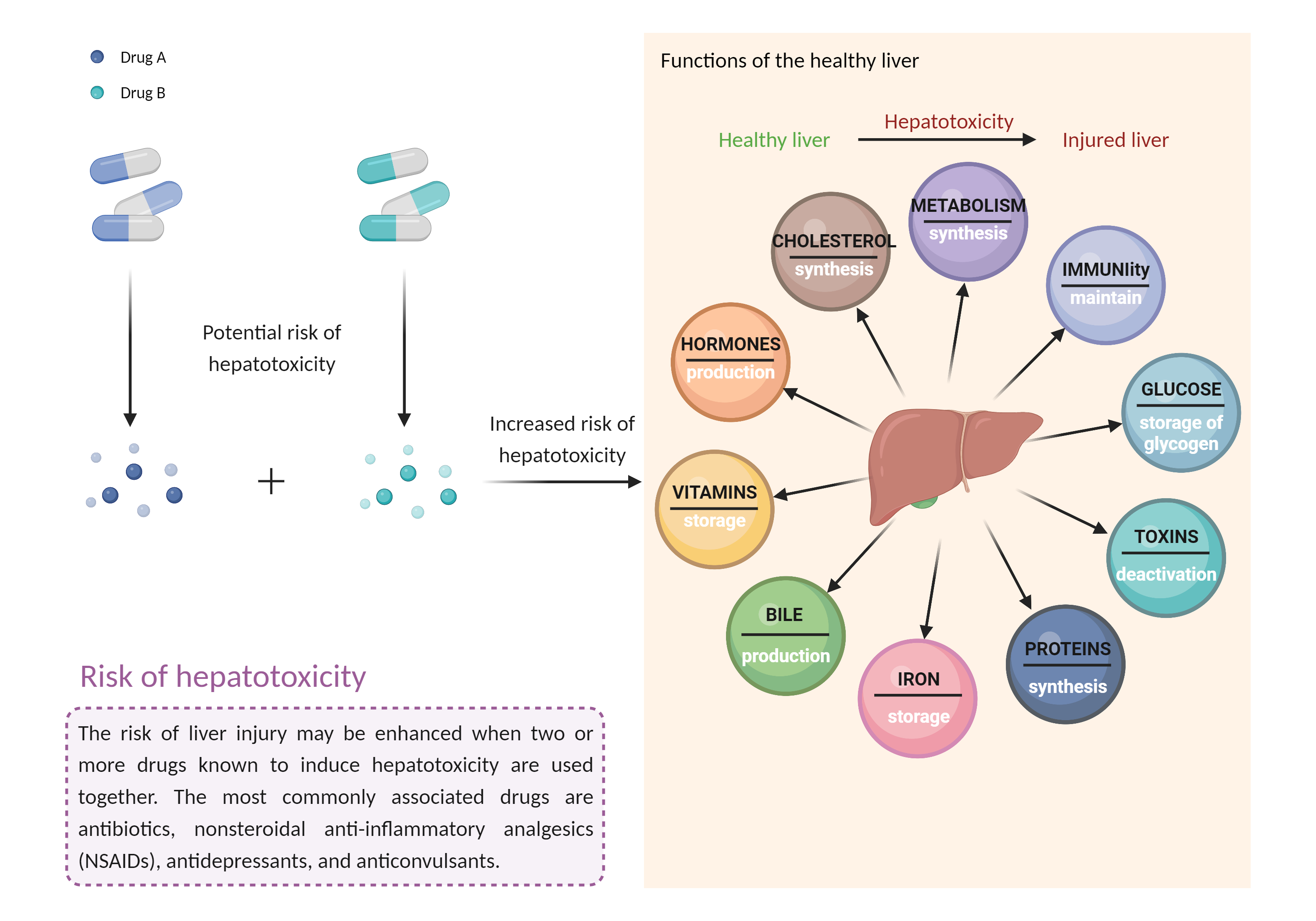
The use of bedaquiline with other potentially hepatotoxic agents should be avoided whenever possible (e.g., acetaminophen alcohol androgens and anabolic steroids other antituberculous agents azole antifungal agents ACE inhibitors disulfiram endothelin receptor antagonists ketolide and macrolide antibiotics interferons kinase inhibitors minocycline nonsteroidal anti-inflammatory agents nucleoside reverse transcriptase inhibitors proteasome inhibitors retinoids sulfonamides tamoxifen thiazolidinediones tolvaptan vincristine zileuton anticonvulsants such as carbamazepine, hydantoins, felbamate, and valproic acid lipid-lowering medications such as fenofibrate, lomitapide, mipomersen, niacin, and statins herbals and nutritional supplements such as black cohosh, chaparral, comfrey, DHEA, kava, pennyroyal oil, and red yeast rice), especially in patients with diminished hepatic reserve. Patients treated with bedaquiline should have serum ALT, AST, alkaline phosphatase, and bilirubin monitored at baseline and monthly during treatment, or as often as needed. An increase of serum aminotransferases to greater than 3 times ULN should be followed by repeat testing within 48 hours. Patients should be advised to seek medical attention if they experience potential signs and symptoms of hepatotoxicity such as fever, rash, itching, anorexia, nausea, vomiting, fatigue, malaise, right upper quadrant pain, dark urine, pale stools, and jaundice. Discontinue bedaquiline if aminotransferase elevations are accompanied by total bilirubin elevation greater than 2 times ULN, aminotransferase elevations are greater than 8 times ULN, or aminotransferase elevations persist beyond 2 weeks. |