Details of Drug-Drug Interaction
| Drug General Information (ID: DDIRXHL8PS) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Fexofenadine | Drug Info | Enasidenib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiallergic Agents | Antineoplastics | |||||||
| Structure | |||||||||
| Mechanism of Fexofenadine-Enasidenib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
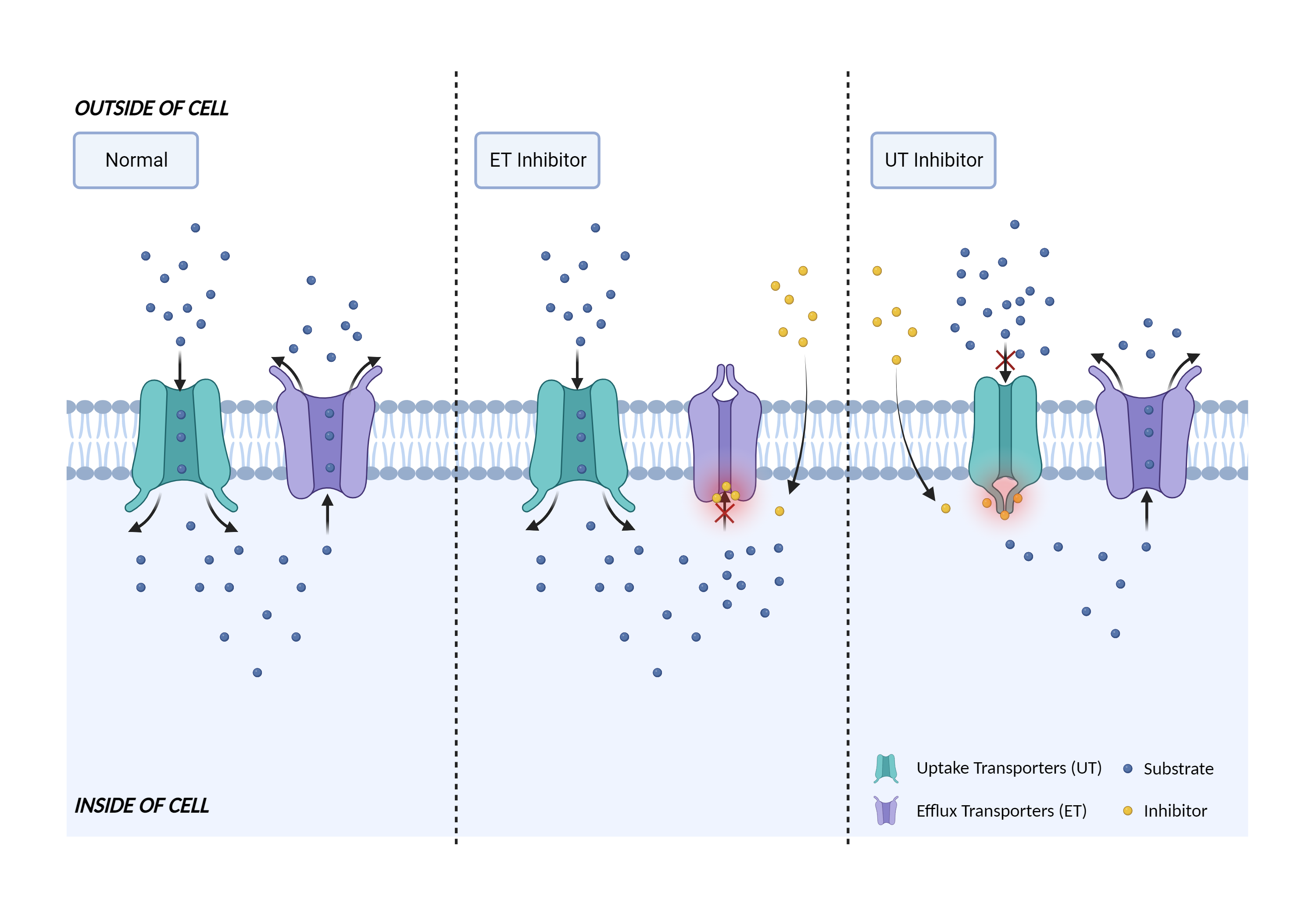
| Transporter inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Fexofenadine | Enasidenib | |||||||
| Mechanism | OATP1B3 substrate | OATP1B3 inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Liver organic anion transporter 2 |
×
Structure
Sequence
MDQHQHLNKTAESASSEKKKTRRCNGFKMFLAALSFSYIAKALGGIIMKISITQIERRFDISSSLAGLIDGSFEIGNLLVIVFVSYFGSKLHRPKLIGIGCLLMGTGSILTSLPHFFMGYYRYSKETHINPSENSTSSLSTCLINQTLSFNGTSPEIVEKDCVKESGSHMWIYVFMGNMLRGIGETPIVPLGISYIDDFAKEGHSSLYLGSLNAIGMIGPVIGFALGSLFAKMYVDIGYVDLSTIRITPKDSRWVGAWWLGFLVSGLFSIISSIPFFFLPKNPNKPQKERKISLSLHVLKTNDDRNQTANLTNQGKNVTKNVTGFFQSLKSILTNPLYVIFLLLTLLQVSSFIGSFTYVFKYMEQQYGQSASHANFLLGIITIPTVATGMFLGGFIIKKFKLSLVGIAKFSFLTSMISFLFQLLYFPLICESKSVAGLTLTYDGNNSVASHVDVPLSYCNSECNCDESQWEPVCGNNGITYLSPCLAGCKSSSGIKKHTVFYNCSCVEVTGLQNRNYSAHLGECPRDNTCTRKFFIYVAIQVINSLFSATGGTTFILLTVKIVQPELKALAMGFQSMVIRTLGGILAPIYFGALIDKTCMKWSTNSCGAQGACRIYNSVFFGRVYLGLSIALRFPALVLYIVFIFAMKKKFQGKDTKASDNERKVMDEANLEFLNNGEHFVPSAGTDSKTCNLDMQDNAAAN
|
|||||||
| Gene Name | OATP1B3 | ||||||||
| Uniprot ID | SO1B3_HUMAN | ||||||||
| KEGG Pathway | hsa:28234 | ||||||||
| Protein Family | Organo anion transporter (TC 2.A.60) family | ||||||||
| Protein Function |
Mediates the Na(+)-independent uptake of organic anions such as 17-beta-glucuronosyl estradiol, taurocholate, triiodothyronine (T3), leukotriene C4, dehydroepiandrosterone sulfate (DHEAS), methotrexate and sulfobromophthalein (BSP). Involved in the clearance of bile acids and organic anions from the liver.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised when enasidenib is used concurrently with medications that are substrates of OATP 1B1/1B3 and/or BCRP, particularly those with a narrow therapeutic range. Dosage adjustments as well as clinical and laboratory monitoring may be appropriate for some drugs whenever enasidenib is added to or withdrawn from therapy. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "Australian Product Information.". | ||||||||||||||||||
| 2 | Product Information. Idhifa (enasidenib). Celgene Corporation, Summit, NJ. | ||||||||||||||||||
