Details of Drug-Drug Interaction
| Drug General Information (ID: DDIR52ZYHK) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Dapsone (topical) | Drug Info | Nitrofurantoin | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiinflammatory Agents | Antiinfective Agents | |||||||
| Structure | |||||||||
| Mechanism of Dapsone (topical)-Nitrofurantoin Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
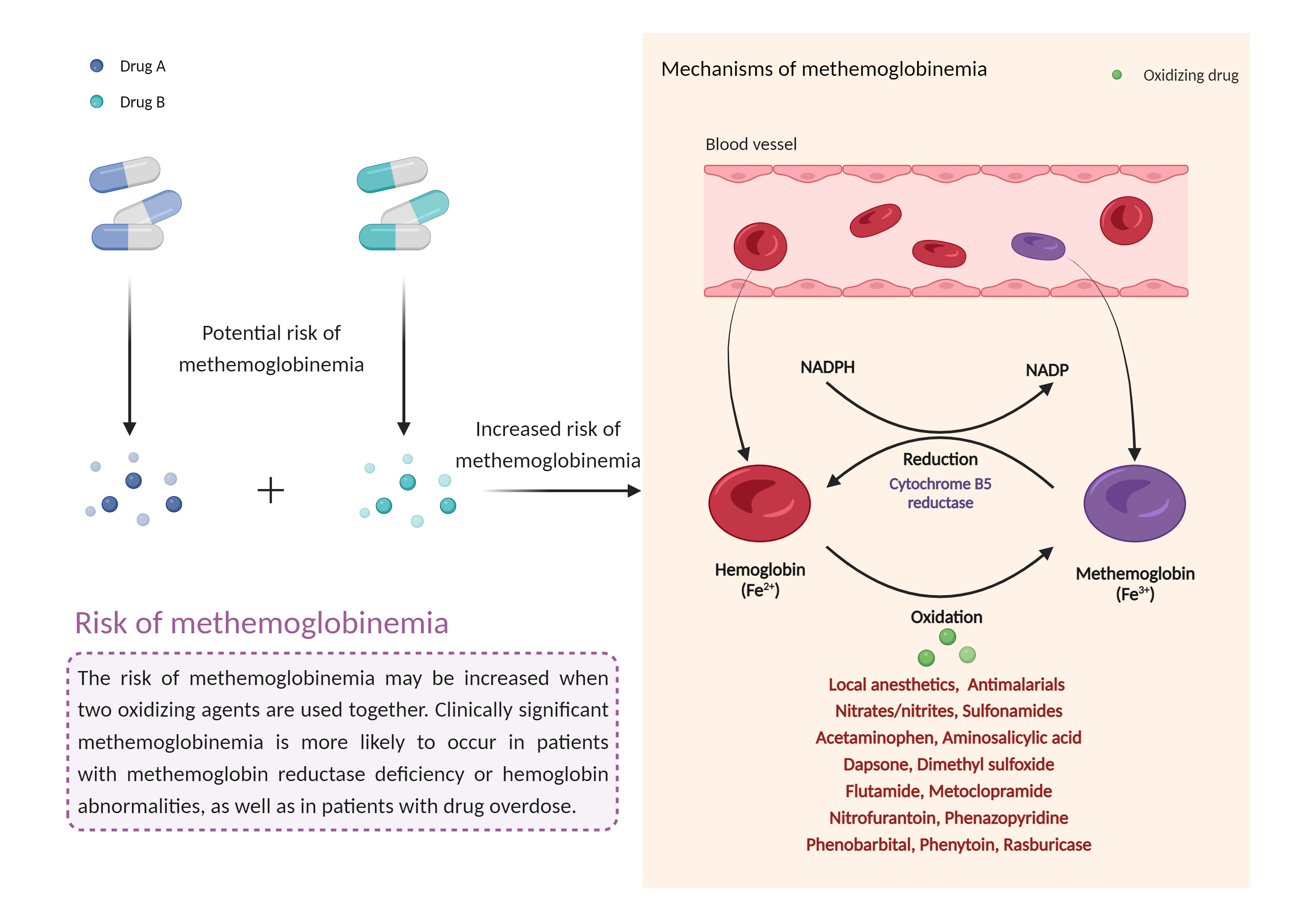
| Increased risk of methemoglobinemia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Dapsone (topical) | Nitrofurantoin | |||||||
| Mechanism | Methemoglobinemia | Methemoglobinemia | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Methemoglobinemia | ||||||||
| Factor Description | Methemoglobinemia is a condition in which higher than normal levels of methemoglobin are found in the blood. Methemoglobinemia is a potentially life-threatening condition in which the oxygen-carrying capacity of circulating hemoglobin is reduced due to the conversion of some or all of the four iron species from the reduced ferrous (Fe2+) state to the oxidized iron (Fe3+) state. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concurrent use of oral or topical dapsone with other methemoglobinemia-inducing medications should be followed with monitoring for signs and symptoms of methemoglobinemia (e.g., cyanosis, hypoxia). | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "Australian Product Information.". | ||||||||||||||||||
| 2 | Product Information. Aczone (dapsone topical). QLT USA, Inc, Fort Collins, CO. | ||||||||||||||||||
