Details of Drug-Drug Interaction
| Drug General Information (ID: DDIQ7LRBVT) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Sevelamer | Drug Info | Mycophenolic acid | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Phosphate Binders | Antiviral Agents | |||||||
| Structure | |||||||||
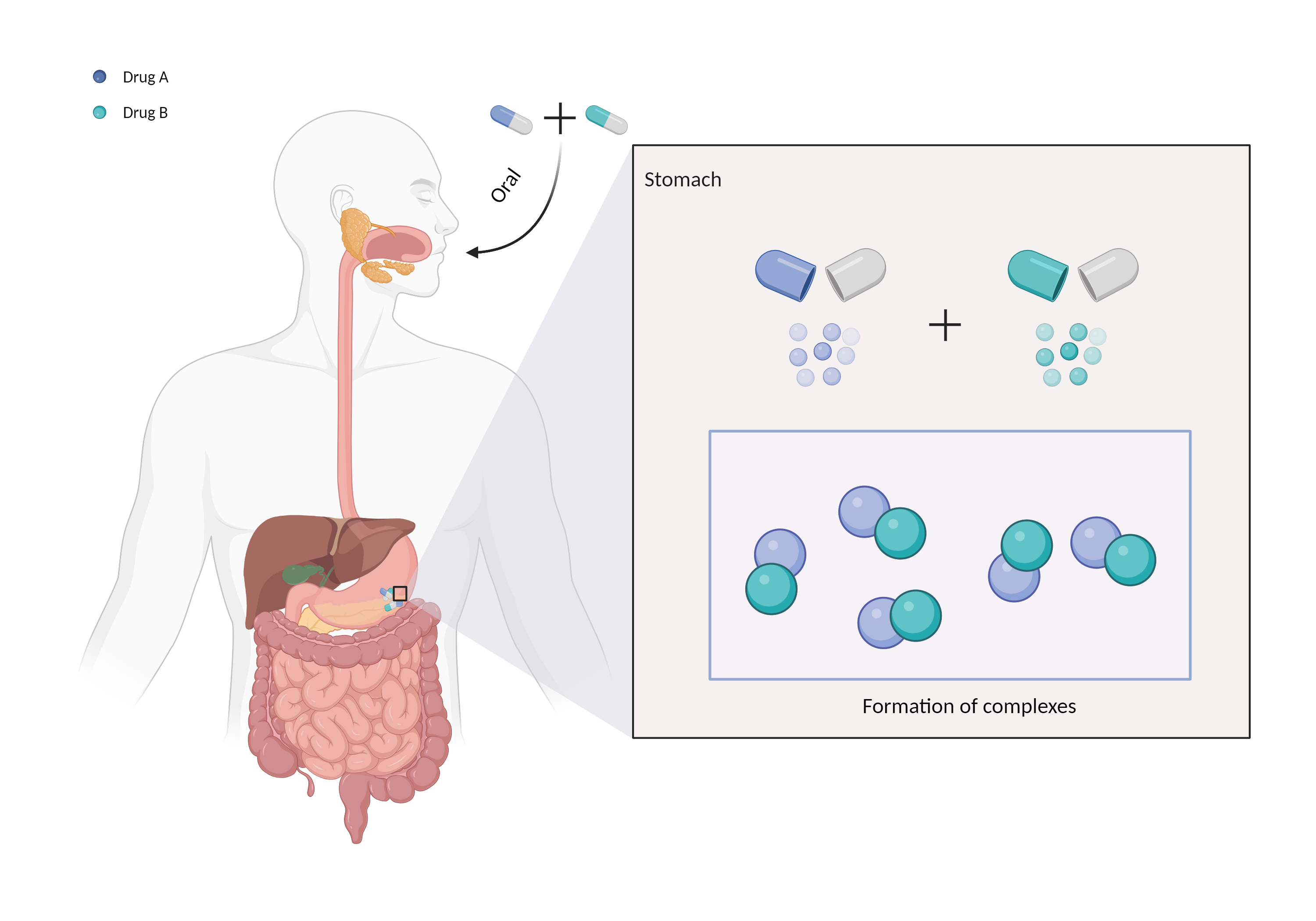
| Mechanism of Sevelamer-Mycophenolic acid Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Complex formation Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Sevelamer | Mycophenolic acid | |||||||
| Mechanism | Anion exchange resin/polymer | Binds to anion exchange resin/polymer | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Chelation | ||||||||
| Factor Description | Chelation is a direct drug interaction that usually involves the formation of dimers or trimers, resulting in larger complexes and poorer absorption. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The immunosuppressant may be given at least 1 to 2 hours before or 2 to 3 hours after sevelamer administration to minimize the impact on absorption. It is recommended to closely monitor blood concentrations of the immunosuppressant during concomitant use and after discontinuation of sevelamer. | ||||||||
