Details of Drug-Drug Interaction
| Drug General Information (ID: DDIPZ8HMVI) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Disulfiram | Drug Info | Abacavir | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Alcohol Deterrents | Anti-Hiv Agents | |||||||
| Structure | |||||||||
| Mechanism of Disulfiram-Abacavir Interaction (Severity Level: Minor) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Competitive inhibition of metabolic enzyme Click to Show/Hide Mechanism Graph | |||||||||
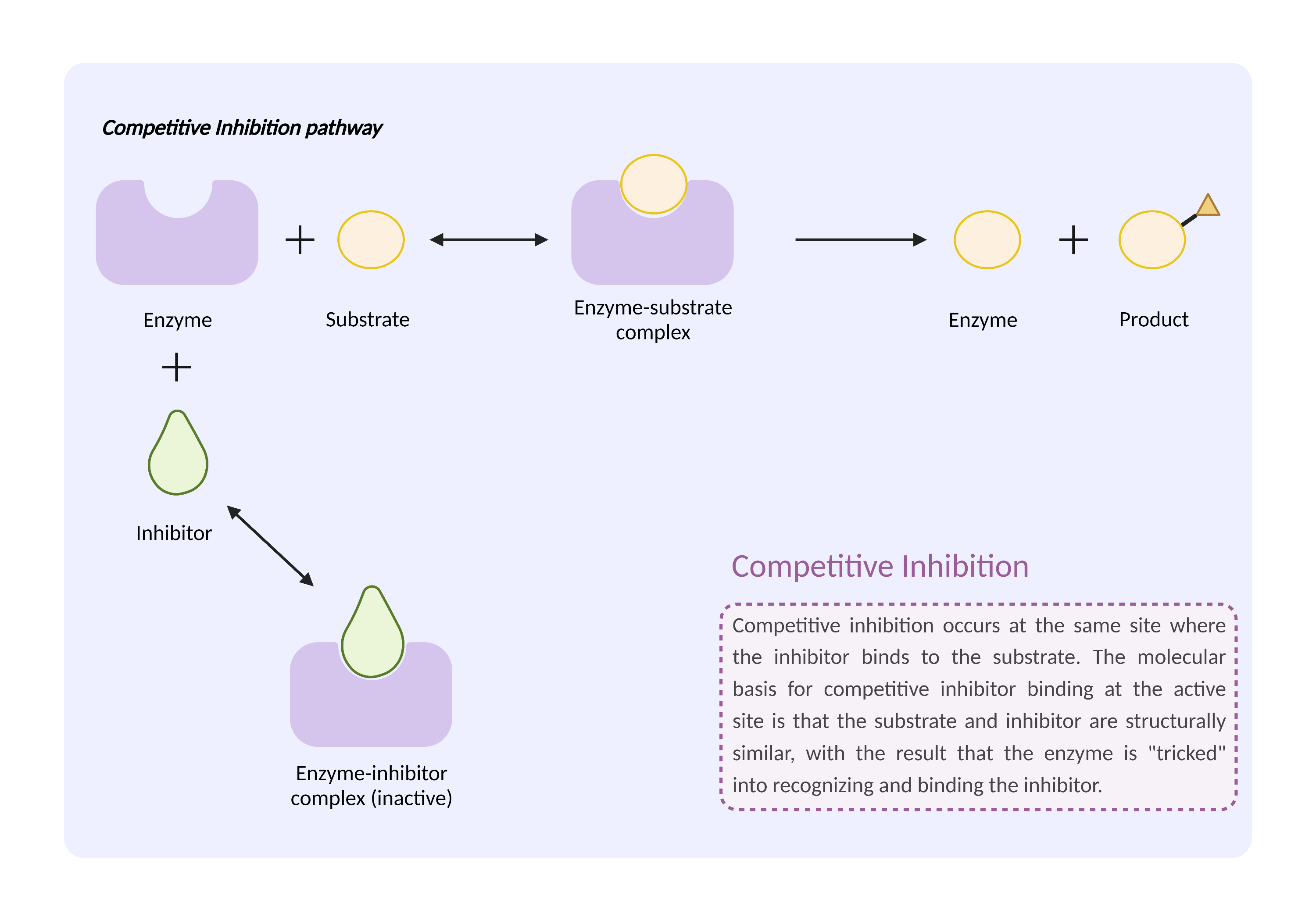 |
|||||||||
| Drug Name | Disulfiram | Abacavir | |||||||
| Mechanism 1 | Alcohol dehydrogenase substrate | Alcohol dehydrogenase substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Alcohol dehydrogenase | Structure Sequence | |||||||
| Protein Family | Zinc-containing alcohol dehydrogenase family | ||||||||
| Protein Function |
Alcohol dehydrogenase (PubMed:2738060). Oxidizes primary as well as secondary alcohols. Ethanol is a very poor substrate (PubMed:2738060).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Mechanism 2 | UGT substrate | UGT substrate | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | UDP-glucuronosyltransferase | Structure Sequence | |||||||
| Protein Family | UDP-glycosyltransferase family | ||||||||
| Protein Function |
[Isoform 1]: UDP-glucuronosyltransferase (UGT) that catalyzes phase II biotransformation reactions in which lipophilic substrates are conjugated with glucuronic acid to increase the metabolite's water solubility, thereby facilitating excretion into either the urine or bile (PubMed:12181437, PubMed:15472229, PubMed:18004206, PubMed:18004212, PubMed:18719240, PubMed:19830808, PubMed:23288867). Essential for the elimination and detoxification of drugs, xenobiotics and endogenous compounds (PubMed:12181437, PubMed:18004206, PubMed:18004212). Catalyzes the glucuronidation of endogenous estrogen hormones such as estradiol, estrone and estriol (PubMed:15472229, PubMed:18719240, PubMed:23288867). Involved in the glucuronidation of bilirubin, a degradation product occurring in the normal catabolic pathway that breaks down heme in vertebrates (PubMed:17187418, PubMed:18004206, PubMed:19830808). Also catalyzes the glucuronidation the isoflavones genistein, daidzein, glycitein, formononetin, biochanin A and prunetin, which are phytoestrogens with anticancer and cardiovascular properties (PubMed:18052087, PubMed:19545173). Involved in the glucuronidation of the AGTR1 angiotensin receptor antagonist losartan, a drug which can inhibit the effect of angiotensin II (PubMed:18674515). Involved in the biotransformation of 7-ethyl-10-hydroxycamptothecin (SN-38), the pharmacologically active metabolite of the anticancer drug irinotecan (PubMed:12181437, PubMed:18004212, PubMed:20610558).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Ziagen (abacavir). Glaxo Wellcome, Research Triangle Pk, NC. | ||||||||||||||||||
