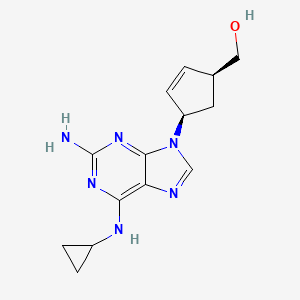
Details of Drug
| Drug-Drug Interaction Network | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sunburst Graph |
AM001 Affected gastrointestinal absorption
AM004 Affected intra/extra-hepatic metabolism
AM006 Pharmacodynamic additive effects
AM007 Pharmacodynamic antagonistic effects
BM009 Altered absorption (Unspecific)
BM017 CYP450 enzyme inhibition
BM018 Non-CYP450 enzyme inhibition
BM024 Competitive inhibition of metabolic enzyme
BM055 Increased risk of hepatotoxicity
BM070 Increased risk of metabolic acidosis
BM107 Attenuated pharmacological effects (Unspecific)
|
||||||||
| Relation Graph | |||||||||
| Full list of drugs interacting with Abacavir | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Affected gastrointestinal absorption | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Altered absorption (Unspecific) | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1187
|
Orlistat
|
C29H53NO5
|
|
Moderate | Inter Info |
[31]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Affected intra/extra-hepatic metabolism | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CYP450 enzyme inhibition | Drug Num: 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0375
|
Cobicistat
|
C40H53N7O5S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1408
|
Riociguat
|
C20H19FN8O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-CYP450 enzyme inhibition | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0617
|
Ethanol
|
C2H6O
|
|
Minor | Inter Info |
[39]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Competitive inhibition of metabolic enzyme | Drug Num: 5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0311
|
Chloral hydrate
|
C2H3Cl3O2
|
|
Minor | Inter Info |
[39]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0324
|
Chlorpromazine
|
C17H19ClN2S
|
|
Minor | Inter Info |
[39]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0327
|
Chlorzoxazone
|
C7H4ClNO2
|
|
Minor | Inter Info |
[39]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0509
|
Disulfiram
|
C10H20N2S4
|
|
Minor | Inter Info |
[39]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0858
|
Isoniazid
|
C6H7N3O
|
|
Minor | Inter Info |
[39]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pharmacodynamic additive effects | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Increased risk of hepatotoxicity | Drug Num: 23 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0900
|
Leflunomide
|
C12H9F3N2O2
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0944
|
Lomitapide
|
C39H37F6N3O2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1073
|
Mipomersen
|
C230H324N67O122P19S19
|
|
Major | Inter Info |
[5]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1258
|
Pexidartinib
|
C20H15ClF3N5
|
|
Major | Inter Info |
[6]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1556
|
Teriflunomide
|
C12H9F3N2O2
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0113
|
Asparaginase Erwinia chrysanthemi
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0199
|
Brentuximab vedotin
|
NA
|
NA
|
Moderate | Inter Info |
[20]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0232
|
Calaspargase pegol
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0248
|
Cannabidiol
|
C21H30O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0789
|
Idelalisib
|
C22H18FN7O
|
|
Moderate | Inter Info |
[30]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1239
|
Peginterferon beta-1a
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1389
|
Remdesivir
|
C27H35N6O8P
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1615
|
Trabectedin
|
C39H43N3O11S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0114
|
Asparaginase Escherichia coli
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0357
|
Clofarabine
|
C10H11ClFN5O3
|
|
Moderate | Inter Info |
[43]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0554
|
Efavirenz
|
C14H9ClF3NO2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0584
|
Epirubicin
|
C27H29NO11
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0826
|
Interferon beta-1a
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0827
|
Interferon beta-1b
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1025
|
Methotrexate
|
C20H22N8O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1106
|
Naltrexone
|
C20H23NO4
|
|
Moderate | Inter Info |
[47]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1234
|
Pegaspargase
|
C14H19NO
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1589
|
Tioguanine
|
C5H5N5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Increased risk of metabolic acidosis | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1397
|
Ribavirin
|
C8H12N4O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pharmacodynamic antagonistic effects | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Attenuated pharmacological effects (Unspecific) | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0348
|
Cladribine
|
C10H12ClN5O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||