| Management |
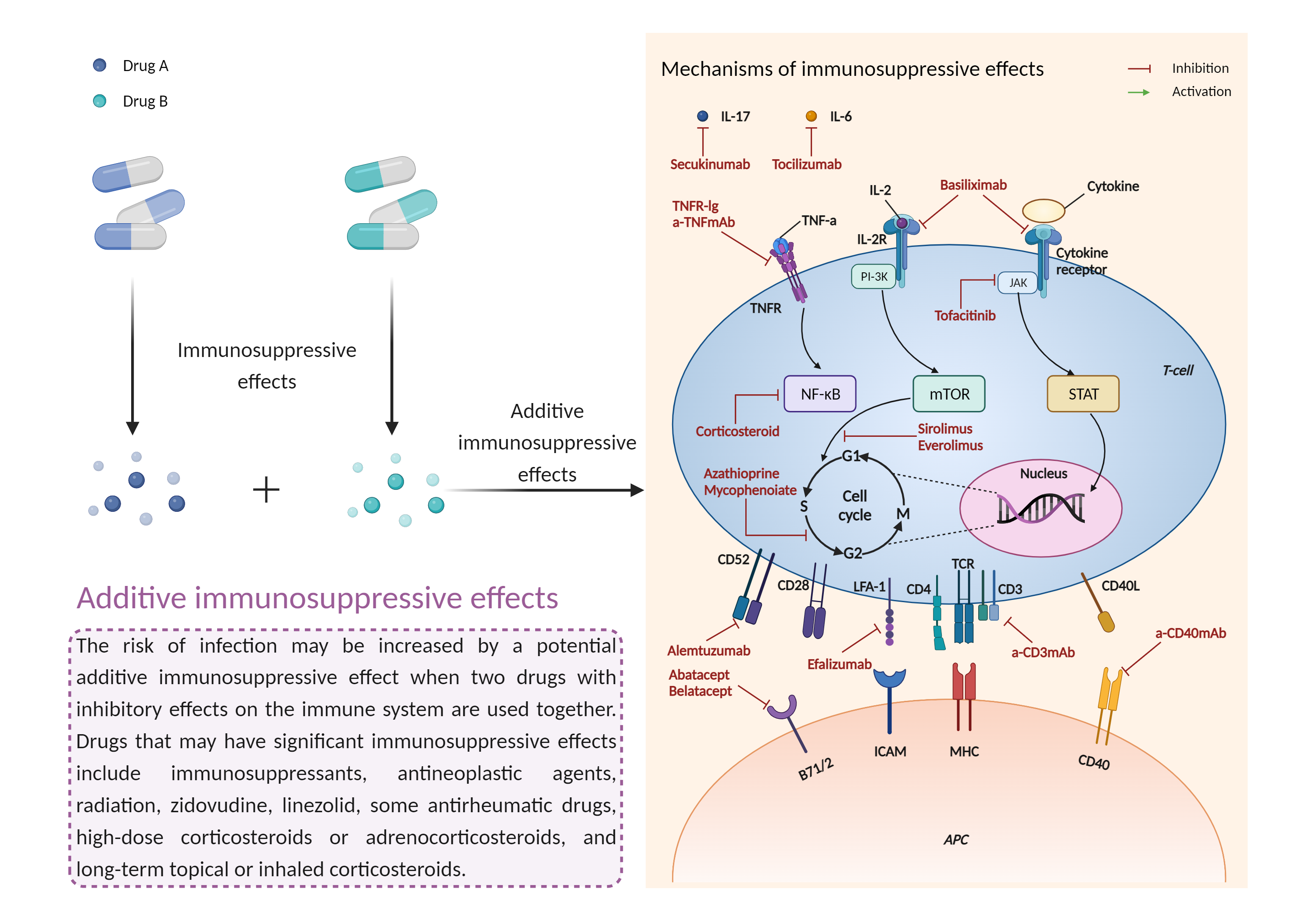
Concomitant use of leflunomide with other immuno- or myelosuppressive antirheumatic agents should generally be avoided. When switching from leflunomide to one of these agents, consideration should be given to administering a washout procedure with cholestyramine or activated charcoal to accelerate elimination of leflunomide's active metabolite from plasma, which otherwise may take up to two years. This will decrease the overlap of systemic exposure to both compounds and reduce the likelihood of additive hematologic toxicities. However, the washout procedure may also induce disease worsening if the patient had been responding to leflunomide treatment. Patients who do not receive the washout procedure prior to switching should be monitored closely for hematologic toxicity. All patients treated with leflunomide should be advised to contact their physician if they develop signs and symptoms of infection such as fever, chills, diarrhea, sore throat, muscle aches, shortness of breath, blood in phlegm, weight loss, red or inflamed skin, body sores, and pain or burning during urination. If evidence of serious infection or bone marrow suppression occurs, treatment with leflunomide should be stopped and washout procedure administered. |