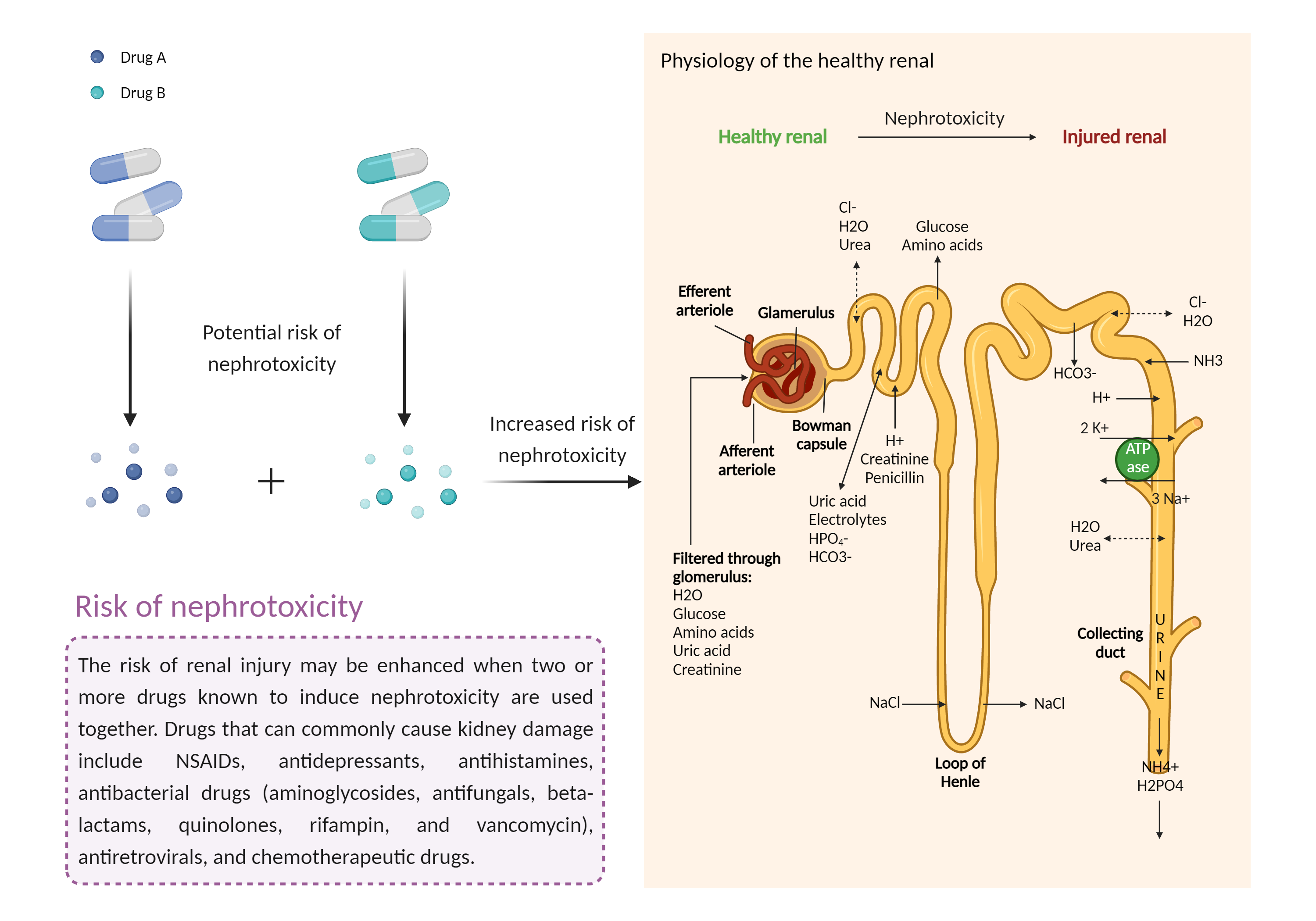
| Management |
Caution is advised when inotersen is prescribed with nephrotoxic drugs and other drugs that may impair renal function (e.g., aminoglycosides polypeptide, glycopeptide, and polymyxin antibiotics amphotericin B aminosalicylates antiviral/antiretroviral agents such as acyclovir, adefovir, cidofovir, foscarnet, ganciclovir, and tenofovir antineoplastics such as aldesleukin, cisplatin, clofarabine, ifosfamide, streptozocin, and high intravenous dosages of methotrexate chelating agents such as deferasirox, deferoxamine, edetate disodium, and edetate calcium disodium immunosuppressants such as cyclosporine, everolimus, sirolimus, and tacrolimus intravascular contrast media intravenous bisphosphonates intravenous pentamidine high dosages and/or chronic use of nonsteroidal anti-inflammatory agents gallium nitrate lithium penicillamine) . Serum creatinine, estimated glomerular filtration rate (eGFR), urine protein to creatinine ratio (UPCR), and a urinalysis should be obtained prior to initiation of inotersen and regularly during and for at least 8 weeks after treatment in accordance with the product labeling. Inotersen should generally not be initiated in patients with a UPCR of 1000 mg/g or higher, or in patients who are unable to adhere to the recommended laboratory monitoring and management guidelines. Patients or their caregivers should be apprised of the signs and symptoms of glomerulonephritis and to seek medical attention if they occur, including edema, shortness of breath, coughing, hematuria, and decreased urination. |