Details of Drug-Drug Interaction
| Drug General Information (ID: DDIODBW4RE) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Theophylline | Drug Info | Riociguat | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Bronchodilators | Vasodilator Agents | |||||||
| Structure | |||||||||
| Mechanism of Theophylline-Riociguat Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
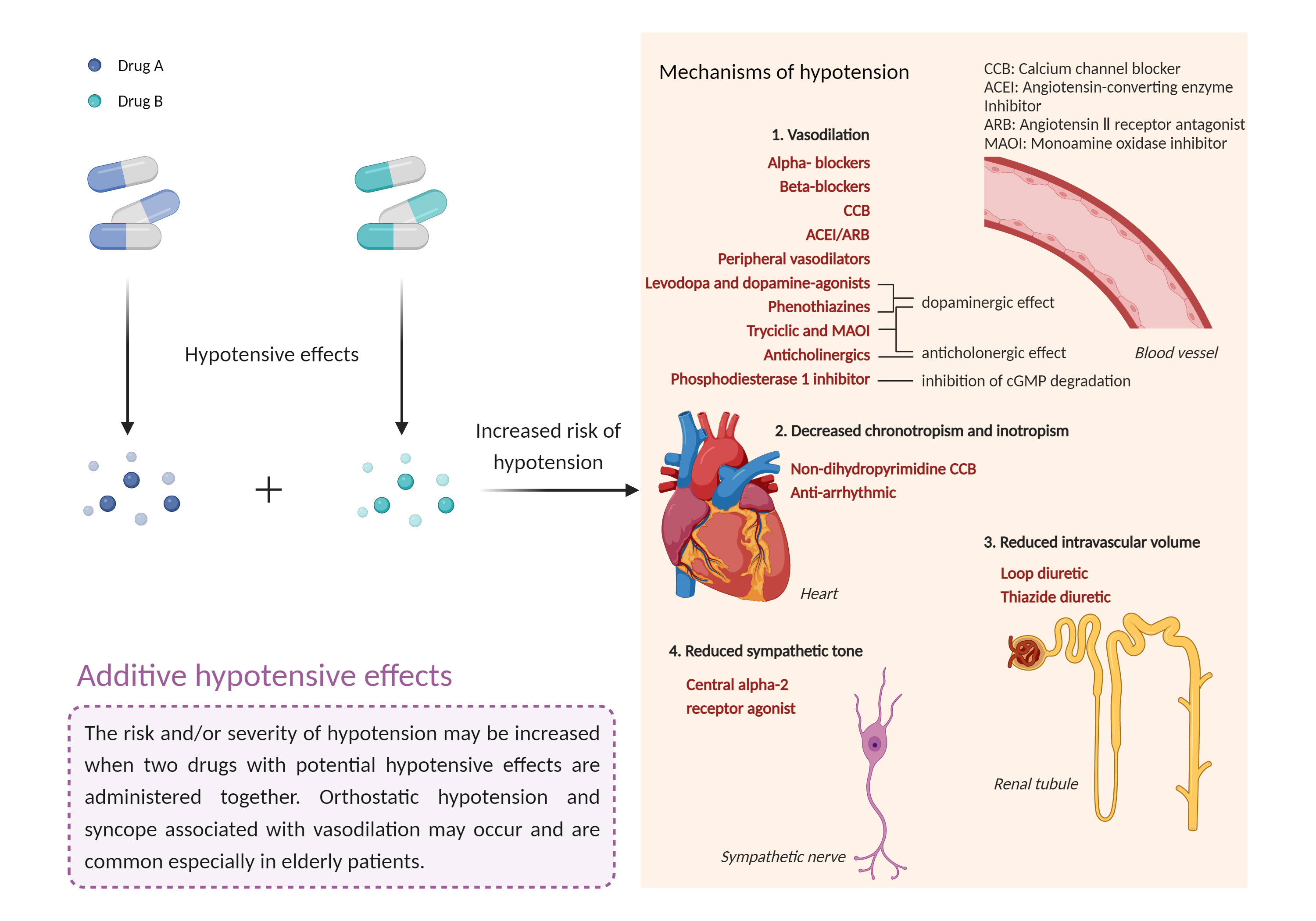 |
|||||||||
| Drug Name | Theophylline | Riociguat | |||||||
| Mechanism |
Hypotensive effects Phosphodiesterase 4 Inhibitor |
Hypotensive effects Soluble guanylyl cyclase Agonist |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Phosphodiesterase 4 | Structure Sequence | |||||||
| Protein Family | Cyclic nucleotide phosphodiesterase family | ||||||||
| Protein Function |
Hydrolyzes the second messenger 3',5'-cyclic AMP (cAMP), which is a key regulator of many important physiological processes.
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Guanylate cyclase soluble | Structure Sequence | |||||||
| Protein Family | Adenylyl cyclase class-4/guanylyl cyclase family | ||||||||
| Protein Function |
Mediates responses to nitric oxide (NO) by catalyzing the biosynthesis of the signaling molecule cGMP.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of riociguat with PDE inhibitors, including specific PDE-5 inhibitors (e.g., avanafil, sildenafil, tadalafil, vardenafil) or nonspecific PDE inhibitors (e.g., dipyridamole, theophylline), is considered contraindicated. Riociguat should be discontinued at least 24 hours before administering a PDE-5 inhibitor, and avoidance of riociguat administration within 24 hours after sildenafil or within 48 hours after tadalafil is recommended. Limited data exists for other PDE inhibitors. | ||||||||
