Details of Drug-Drug Interaction
| Drug General Information (ID: DDIKTLCZJF) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Pravastatin | Drug Info | Paritaprevir | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Statins/Antihyperlipidemic Agents | Antiviral Agents | |||||||
| Structure | |||||||||
| Mechanism of Pravastatin-Paritaprevir Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Transporter inhibition Click to Show/Hide Mechanism Graph | |||||||||
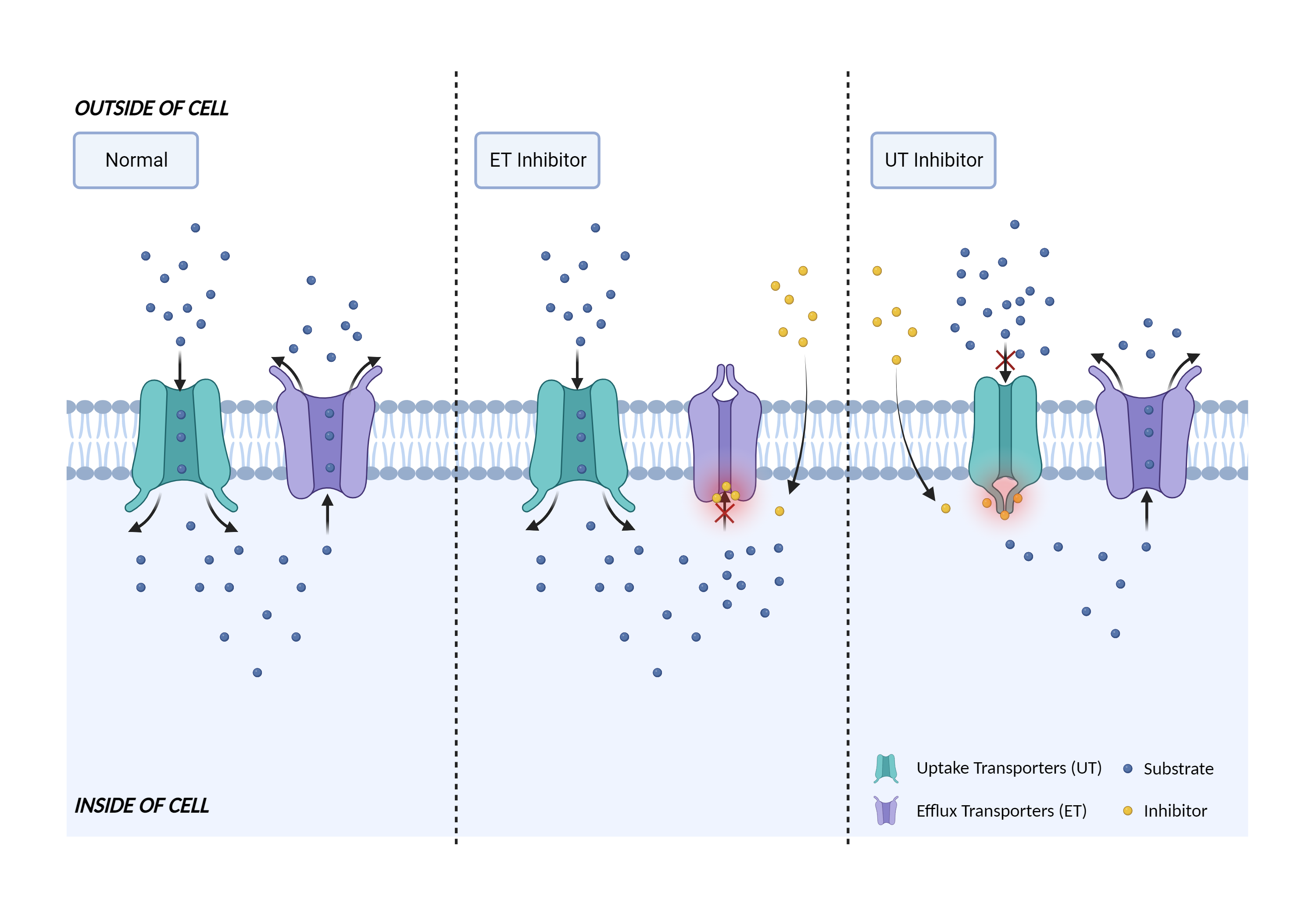 |
|||||||||
| Drug Name | Pravastatin | Paritaprevir | |||||||
| Mechanism | OATP1B1 substrate | OATP1B1 inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Liver organic anion transporter 1 |
×
Structure
Sequence
MDQNQHLNKTAEAQPSENKKTRYCNGLKMFLAALSLSFIAKTLGAIIMKSSIIHIERRFEISSSLVGFIDGSFEIGNLLVIVFVSYFGSKLHRPKLIGIGCFIMGIGGVLTALPHFFMGYYRYSKETNINSSENSTSTLSTCLINQILSLNRASPEIVGKGCLKESGSYMWIYVFMGNMLRGIGETPIVPLGLSYIDDFAKEGHSSLYLGILNAIAMIGPIIGFTLGSLFSKMYVDIGYVDLSTIRITPTDSRWVGAWWLNFLVSGLFSIISSIPFFFLPQTPNKPQKERKASLSLHVLETNDEKDQTANLTNQGKNITKNVTGFFQSFKSILTNPLYVMFVLLTLLQVSSYIGAFTYVFKYVEQQYGQPSSKANILLGVITIPIFASGMFLGGYIIKKFKLNTVGIAKFSCFTAVMSLSFYLLYFFILCENKSVAGLTMTYDGNNPVTSHRDVPLSYCNSDCNCDESQWEPVCGNNGITYISPCLAGCKSSSGNKKPIVFYNCSCLEVTGLQNRNYSAHLGECPRDDACTRKFYFFVAIQVLNLFFSALGGTSHVMLIVKIVQPELKSLALGFHSMVIRALGGILAPIYFGALIDTTCIKWSTNNCGTRGSCRTYNSTSFSRVYLGLSSMLRVSSLVLYIILIYAMKKKYQEKDINASENGSVMDEANLESLNKNKHFVPSAGADSETHC
|
|||||||
| Gene Name | OATP1B1 | ||||||||
| Uniprot ID | SO1B1_HUMAN | ||||||||
| KEGG Pathway | hsa:10599 | ||||||||
| Protein Family | Organo anion transporter (TC 2.A.60) family | ||||||||
| Protein Function |
Mediates the Na(+)-independent uptake of organic anions such as pravastatin, taurocholate, methotrexate, dehydroepiandrosterone sulfate, 17-beta-glucuronosyl estradiol, estrone sulfate, prostaglandin E2, thromboxane B2, leukotriene C3, leukotriene E4, thyroxine and triiodothyronine. Involved in the clearance of bile acids and organic anions from the liver.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The benefits of using pravastatin with ombitasvir/paritaprevir/ritonavir plus dasabuvir should be carefully weighed against the potentially increased risk of myopathy including rhabdomyolysis. If coadministration is required, the lowest effective dosage of pravastatin should be used and close monitoring for musculoskeletal toxicity is recommended. The dosage of pravastatin should be limited to 40 mg/day. alternatively, an interruption of pravastatin may be considered during treatment with ombitasvir/paritaprevir/ritonavir plus dasabuvir, or a different HMG-CoA reductase inhibitor such as fluvastatin or pitavastatin may be substituted. All patients receiving statin therapy should be advised to promptly report any unexplained muscle pain, tenderness or weakness, particularly if accompanied by fever, malaise and/or dark colored urine. Therapy should be discontinued if creatine kinase is markedly elevated in the absence of strenuous exercise or if myopathy is otherwise suspected or diagnosed. | ||||||||
