Details of Drug-Drug Interaction
| Drug General Information (ID: DDIKIXEJNA) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Aldesleukin | Drug Info | Iohexol | Drug Info | |||||
| Drug Type | Interferons | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Non-Ionic Iodinated Contrast Media | |||||||
| Mechanism of Aldesleukin-Iohexol Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
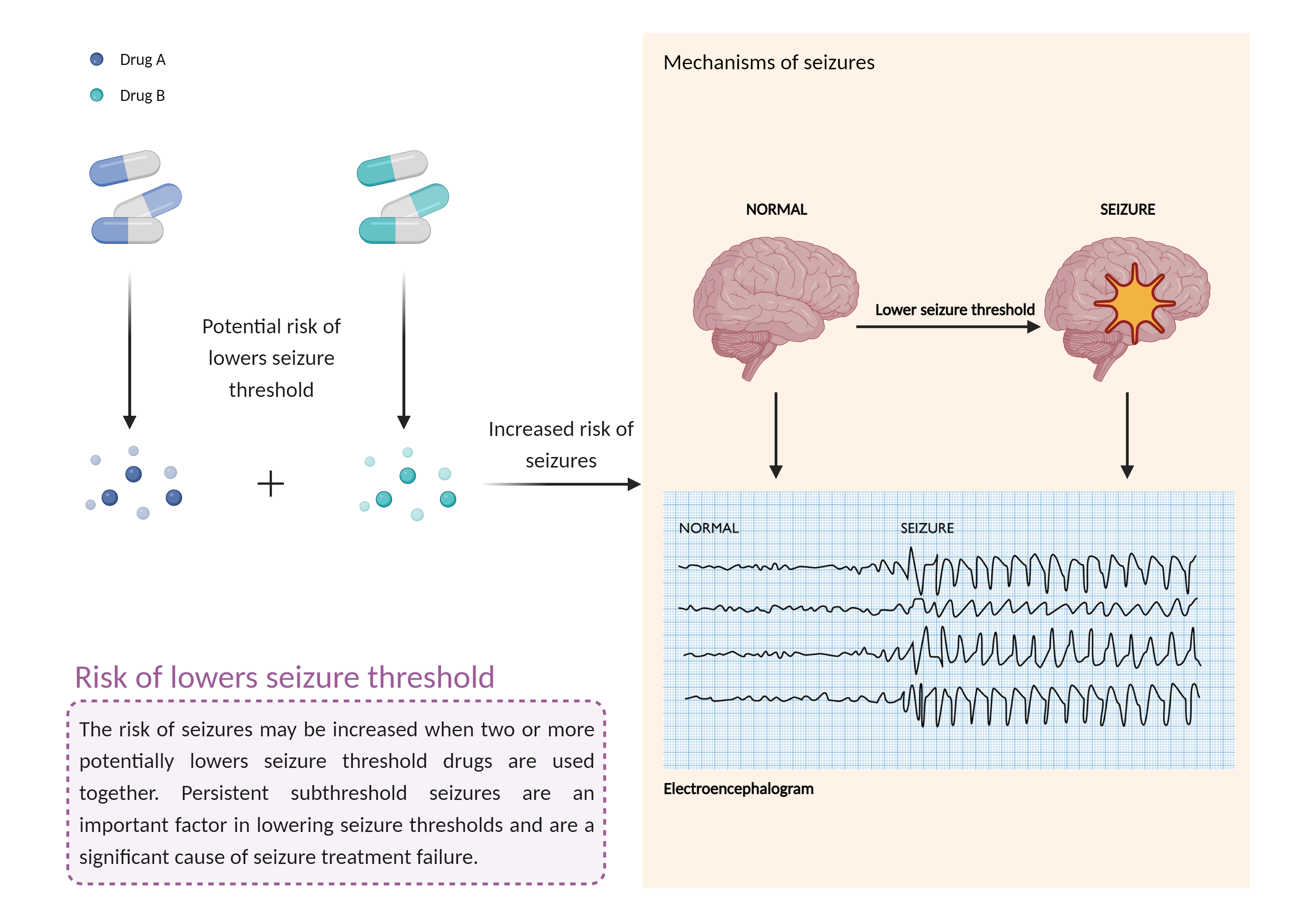
| Increased risk of lowers seizure threshold Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Aldesleukin | Iohexol | |||||||
| Mechanism | Lower seizure threshold | Lower seizure threshold | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Lowers seizure threshold | ||||||||
| Factor Description | The combination of medications that lower the seizure threshold is a factor that makes people with epilepsy more likely to have seizures. A seizure is a sudden, uncontrolled electrical disturbance in the brain that can cause changes in your behavior, movements or sensations, and level of consciousness. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Aldesleukin should preferably be withheld for at least 48 hours prior to and 24 hours following intrathecal administration of iodinated contrast media. Patients should be monitored for adverse reactions during and after the myelography, and symptomatic treatment initiated as clinically appropriate. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Osmovist 240 (iotrolan). Berlex Canada Inc, Lachine, QC. | ||||||||||||||||||
| 2 | Product Information. Proleukin (aldesleukin). Chiron Therapeutics, Emeryville, CA. | ||||||||||||||||||
