Details of Drug-Drug Interaction
| Drug General Information (ID: DDIJFLVMQB) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Bortezomib | Drug Info | Trastuzumab emtansine | Drug Info | |||||
| Drug Type | Small molecule | Monoclonal antibody | |||||||
| Therapeutic Class | Antineoplastics | Antineoplastics/Her2 Inhibitors | |||||||
| Mechanism of Bortezomib-Trastuzumab emtansine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
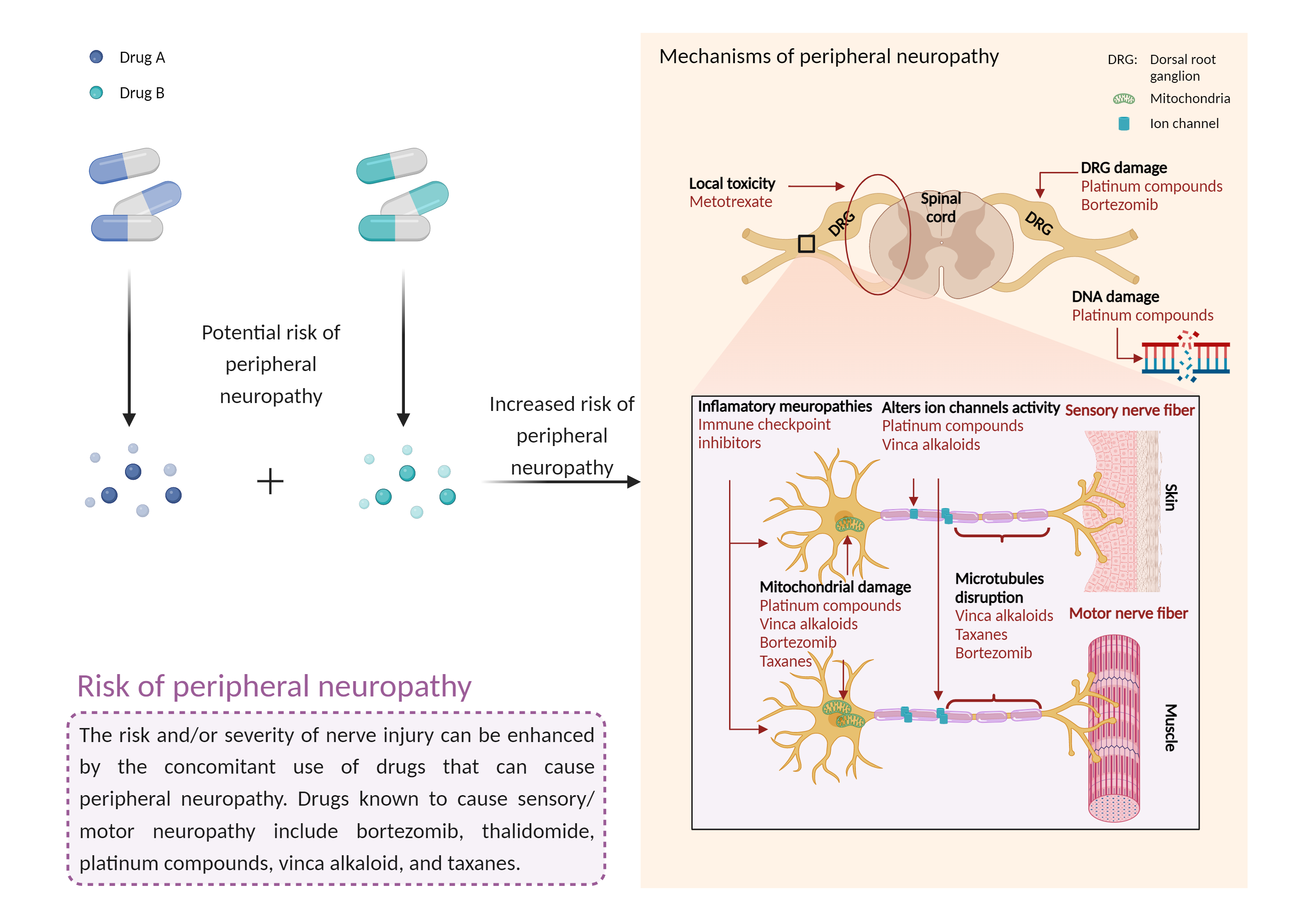
| Increased risk of peripheral neuropathy Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Bortezomib | Trastuzumab emtansine | |||||||
| Mechanism | Peripheral neuropathy | Peripheral neuropathy | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Peripheral neuropathy | ||||||||
| Factor Description | Peripheral neuropathy is a disorder of the peripheral nerves that can cause sensory and motor symptoms, and even paralysis in severe cases. Signs and symptoms of peripheral neuropathy may include: gradual numbness, tingling or prickling in the feet or hands that can spread up the legs and arms; sharp, stinging, pricking or burning sensations; extreme sensitivity to touch; pain during activities that should not cause pain; lack of coordination and falls; muscle weakness; and paralysis if the motor nerves are affected. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if bortezomib is used with other neurotoxic agents. Patients should be closely monitored for symptoms of neuropathy such as visual disturbances or burning, tingling, pain, numbness, and/or weakness in the extremities. Patients experiencing new or worsening peripheral neuropathy may require an adjustment in the dosage and schedule of bortezomib in accordance with the product labeling. | ||||||||
