Details of Drug-Drug Interaction
| Drug General Information (ID: DDIDRVK17X) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Vorapaxar | Drug Info | Dextran (-1) | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiplatelet Agents/Protease-Activated Receptor-1 Antagonists | Plasma Expanders | |||||||
| Structure | |||||||||
| Mechanism of Vorapaxar-Dextran (-1) Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
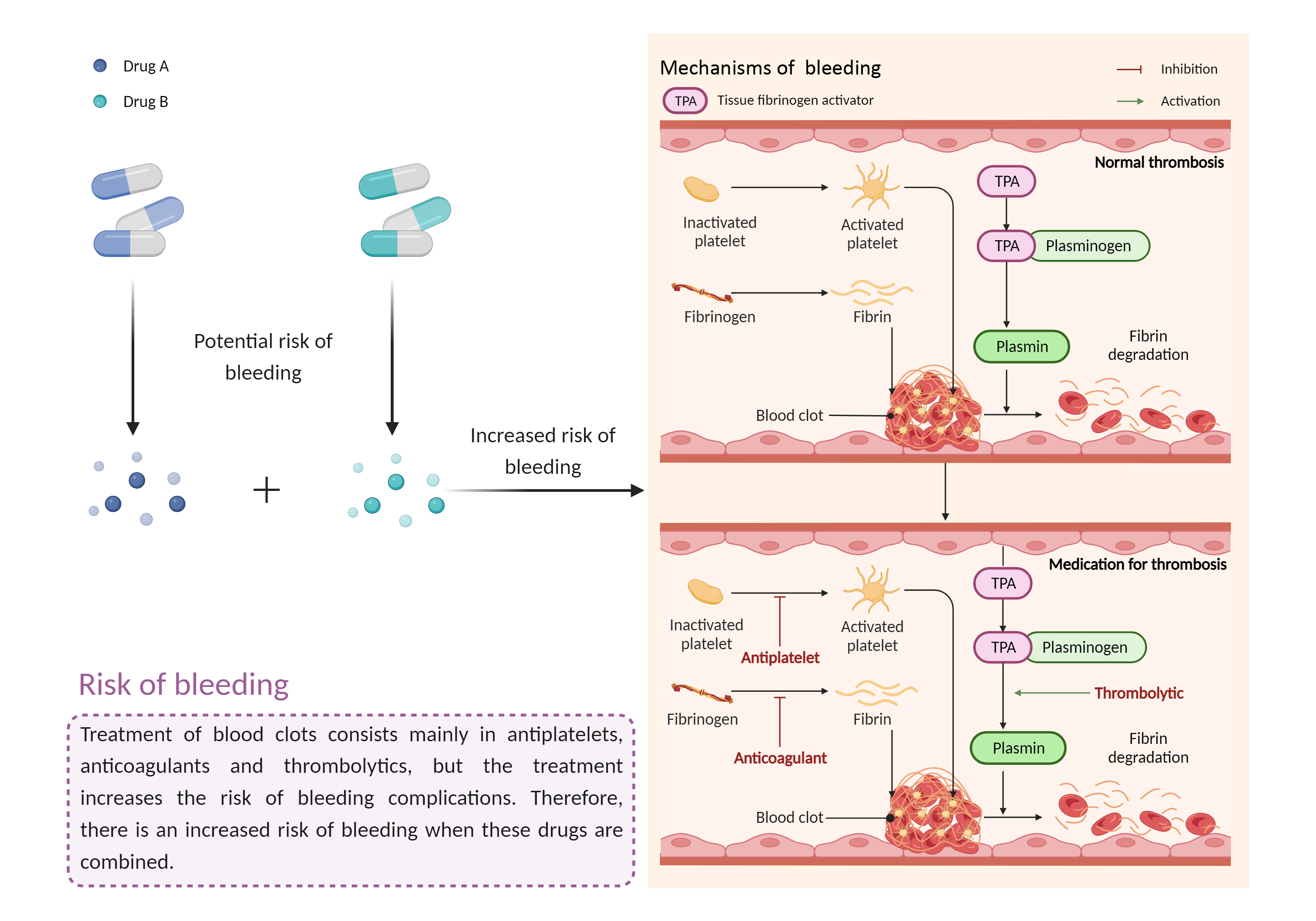
| Increased risk of bleeding Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Vorapaxar | Dextran (-1) | |||||||
| Mechanism |
Risk of bleeding Antiplatelet |
Risk of bleeding Anticoagulant effects |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Bleeding | ||||||||
| Factor Description | Patients may be at risk for bleeding when the outcome of a disease or disease treatment confounds the standard mechanisms for maintaining hemostasis. Signs or symptoms of abnormal bleeding include: bleeding that takes a long time to stop (including nosebleeds, bleeding gums, bleeding from cuts and abrasions, and menstrual bleeding); severe unexplained bruising, or bruising that becomes larger; blood in the urine or stool, etc. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if vorapaxar is used in combination with other drugs that affect hemostasis. Close clinical and laboratory observation for hemorrhagic complications is recommended. Patients should be advised to promptly report any signs of bleeding to their doctor, including pain, swelling, headache, dizziness, weakness, prolonged bleeding from cuts, increased menstrual flow, vaginal bleeding, nosebleeds, bleeding of gums from brushing, unusual bleeding or bruising, red or brown urine, or red or black stools. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Zontivity (vorapaxar). Merck & Company Inc, Whitehouse Station, NJ. | ||||||||||||||||||
