Details of Drug-Drug Interaction
| Drug General Information (ID: DDIABYTJ70) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Trastuzumab emtansine | Drug Info | Inotersen | Drug Info | |||||
| Drug Type | Monoclonal antibody | Small molecule | |||||||
| Therapeutic Class | Antineoplastics/Her2 Inhibitors | Metabolic Agents | |||||||
| Mechanism of Trastuzumab emtansine-Inotersen Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
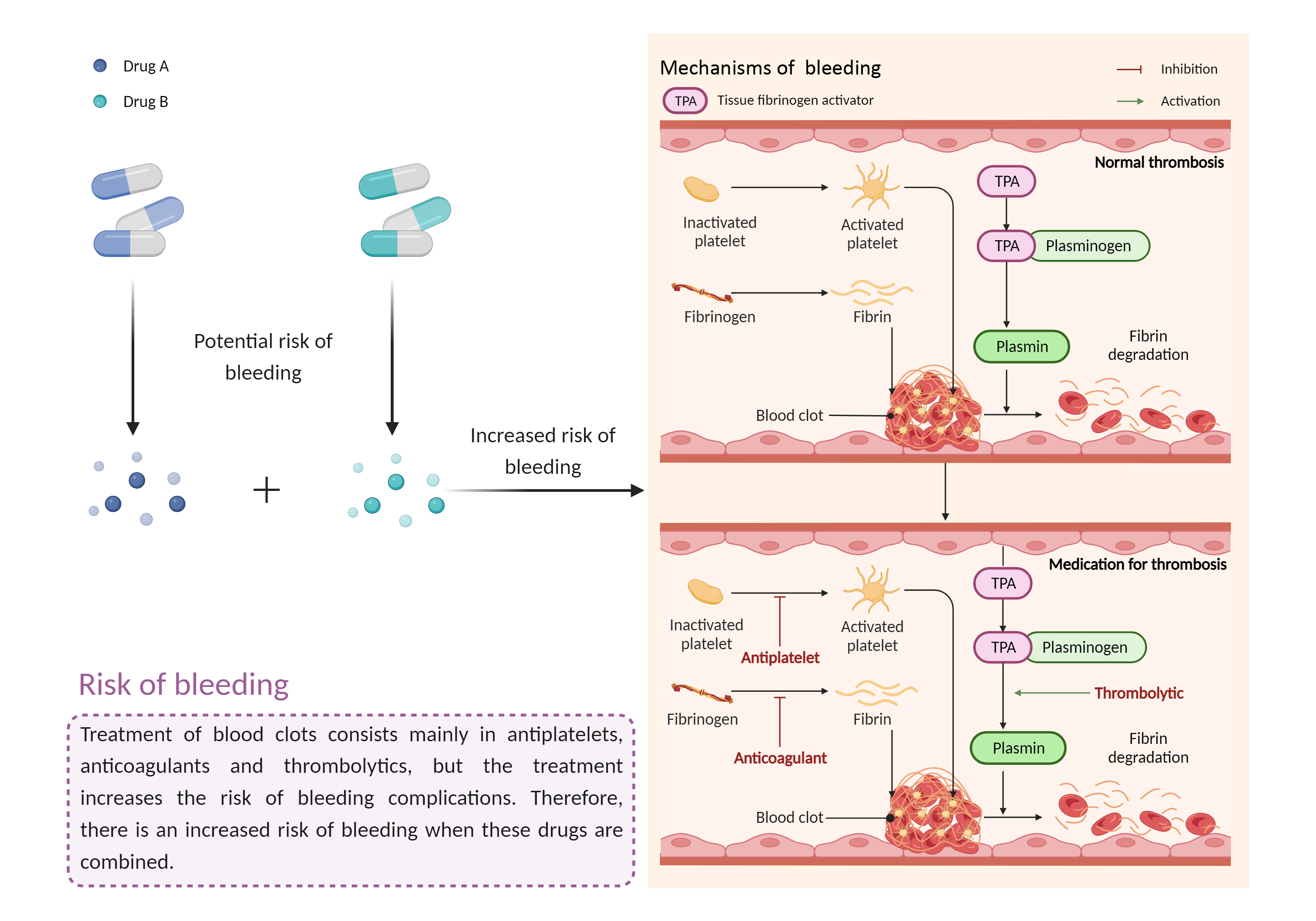
| Increased risk of bleeding Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Trastuzumab emtansine | Inotersen | |||||||
| Mechanism |
Risk of bleeding Antiplatelet effects |
Risk of bleeding Antiplatelet effects |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Bleeding | ||||||||
| Factor Description | Patients may be at risk for bleeding when the outcome of a disease or disease treatment confounds the standard mechanisms for maintaining hemostasis. Signs or symptoms of abnormal bleeding include: bleeding that takes a long time to stop (including nosebleeds, bleeding gums, bleeding from cuts and abrasions, and menstrual bleeding); severe unexplained bruising, or bruising that becomes larger; blood in the urine or stool, etc. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised when inotersen is prescribed with antiplatelet agents, anticoagulants, or other medications that commonly cause thrombocytopenia or bleeding. A platelet count should be obtained prior to initiation of inotersen and regularly during and for at least 8 weeks after treatment in accordance with the product labeling. Inotersen should not be administered in patients with a platelet count below 100 x 10^9/L or in patients who are unable to adhere to the recommended laboratory monitoring and management guidelines. Patients or their caregivers should be apprised of the signs and symptoms of thrombocytopenia and to seek medical attention if they occur, including any unusual or prolonged bleeding (e.g., petechiae, easy bruising, hematoma, subconjunctival bleeding, gingival bleeding, epistaxis, hemoptysis, irregular or heavier than normal menstrual bleeding, hematemesis, hematuria, hematochezia, melena), neck stiffness, or atypical severe headache. | ||||||||
