Details of Drug-Drug Interaction
| Drug General Information (ID: DDI4J7MX5O) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Propofol | Drug Info | Lorlatinib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anxiolytics/Sedatives/Hypnotics | Multikinase Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Propofol-Lorlatinib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
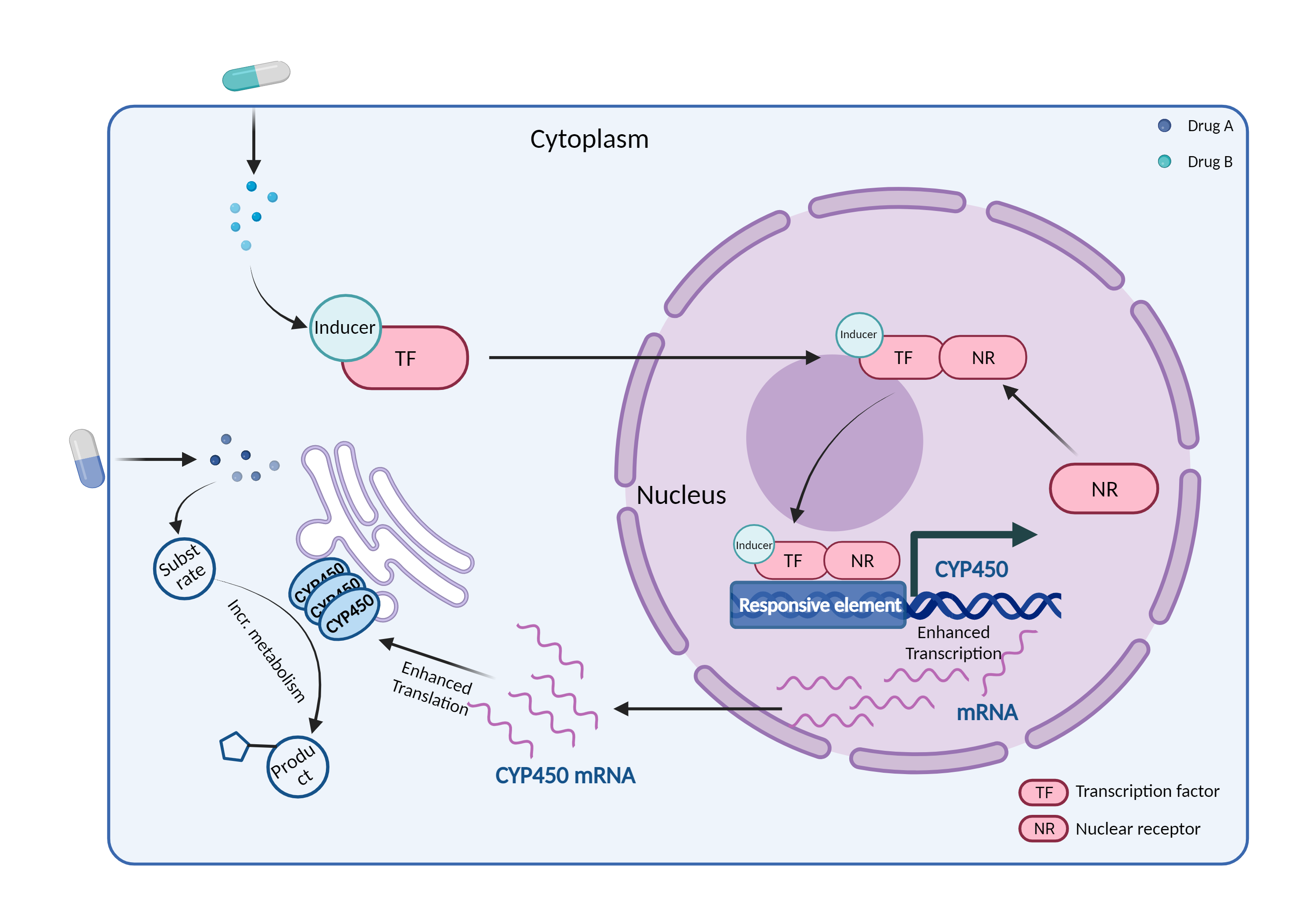
| CYP450 enzyme induction Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Propofol | Lorlatinib | |||||||
| Mechanism | CYP450 2B6 substrate | CYP450 2B6 inducer | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2B6 |
×
Structure
Sequence
MELSVLLFLALLTGLLLLLVQRHPNTHDRLPPGPRPLPLLGNLLQMDRRGLLKSFLRFREKYGDVFTVHLGPRPVVMLCGVEAIREALVDKAEAFSGRGKIAMVDPFFRGYGVIFANGNRWKVLRRFSVTTMRDFGMGKRSVEERIQEEAQCLIEELRKSKGALMDPTFLFQSITANIICSIVFGKRFHYQDQEFLKMLNLFYQTFSLISSVFGQLFELFSGFLKYFPGAHRQVYKNLQEINAYIGHSVEKHRETLDPSAPKDLIDTYLLHMEKEKSNAHSEFSHQNLNLNTLSLFFAGTETTSTTLRYGFLLMLKYPHVAERVYREIEQVIGPHRPPELHDRAKMPYTEAVIYEIQRFSDLLPMGVPHIVTQHTSFRGYIIPKDTEVFLILSTALHDPHYFEKPDAFNPDHFLDANGALKKTEAFIPFSLGKRICLGEGIARAELFLFFTTILQNFSMASPVAPEDIDLTPQECGVGKIPPTYQIRFLPR
|
|||||||
| Gene Name | CYP2B6 | ||||||||
| Uniprot ID | CP2B6_HUMAN | ||||||||
| KEGG Pathway | hsa:1555 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of endocannabinoids and steroids (PubMed:21289075, PubMed:12865317). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase). Catalyzes the epoxidation of double bonds of arachidonoylethanolamide (anandamide) to 8,9-, 11,12-, and 14,15-epoxyeicosatrienoic acid ethanolamides (EpETrE-EAs), potentially modulating endocannabinoid system signaling (PubMed:21289075). Hydroxylates steroid hormones, including testosterone at C-16 and estrogens at C-2 (PubMed:21289075, PubMed:12865317). Plays a role in the oxidative metabolism of xenobiotics, including plant lipids and drugs (PubMed:11695850, PubMed:22909231). Acts as a 1,4-cineole 2-exo-monooxygenase (PubMed:11695850).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution and monitoring are recommended if lorlatinib is used concomitantly with drugs that are substrates of CYP450 2B6, particularly those with a narrow therapeutic range. The potential for diminished therapeutic effects should be considered when lorlatinib is prescribed in combination with drugs that are CYP450 2B6 substrates. However, some authorities suggest that no dose adjustment is necessary when lorlatinib is used with drugs that are substrates of CYP450 2B6 (UK). | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
