Details of Drug-Drug Interaction
| Drug General Information (ID: DDI3J1QOD9) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Magnesium oxide | Drug Info | Cabotegravir | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Minerals And Electrolytes | Antiviral Agents | |||||||
| Structure | |||||||||
| Mechanism of Magnesium oxide-Cabotegravir Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
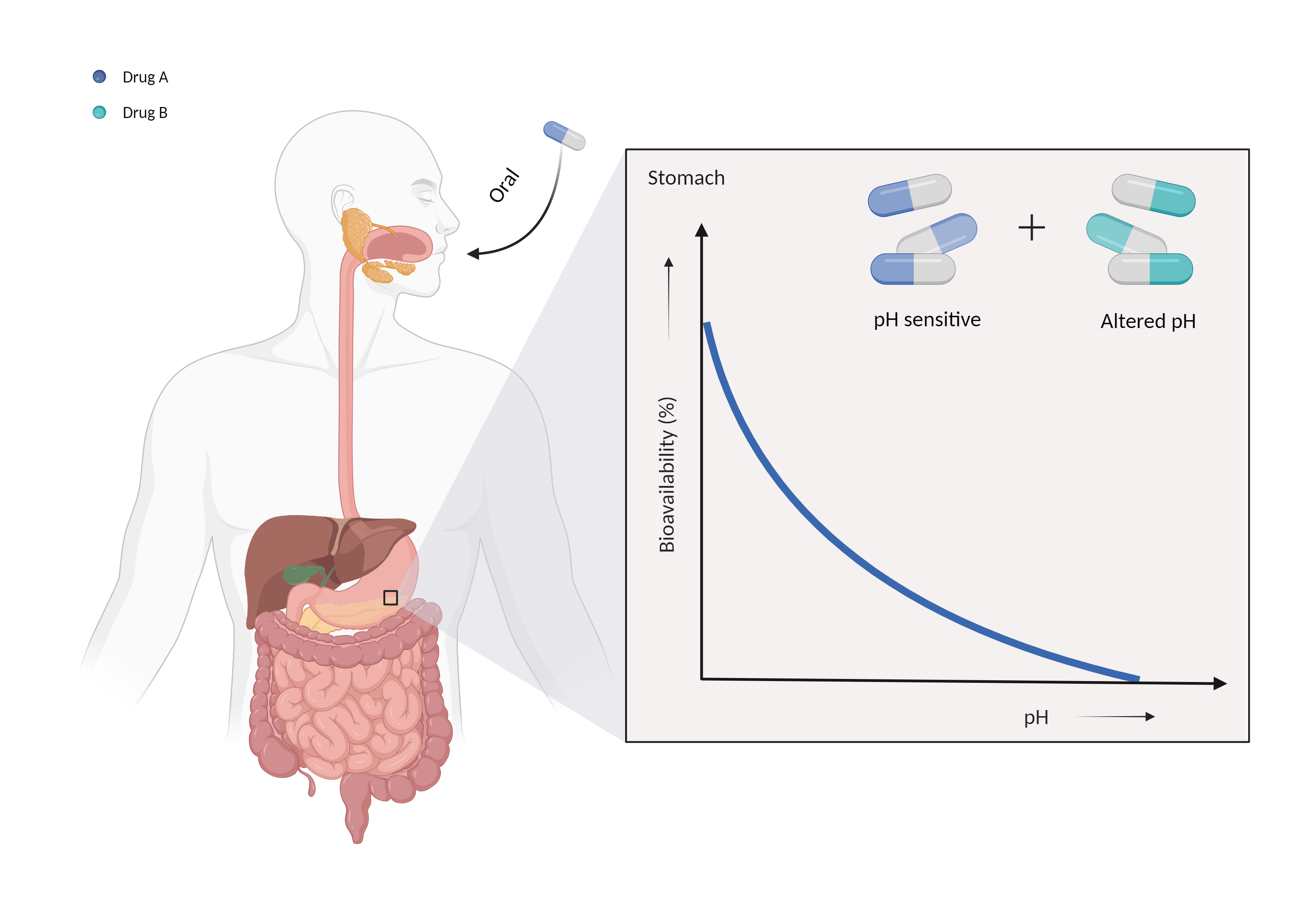
| Altered gastric pH Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Magnesium oxide | Cabotegravir | |||||||
| Mechanism | Gastric alkalinizer | Gastric pH sensitive | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastric pH | ||||||||
| Factor Description | The normal pH range of gastric acid is between 1.5 and 3.5 and is highly acidic, consisting mainly of hydrochloric acid. Changes in the pH of the stomach can alter the absorption of drugs. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | To minimize the risk of reduced viral susceptibility and resistance development associated with subtherapeutic levels of cabotegravir, antacids or other medications that contain antacids (e.g., didanosine buffered tablets or pediatric oral solution) should be administered at least 2 hours before or 4 hours after the oral cabotegravir dose. | ||||||||
