Details of Drug-Drug Interaction
| Drug General Information (ID: DDI2PMDVBC) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Pergolide | Drug Info | Amisulpride | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiparkinson Agents | Antipsychotic Agents | |||||||
| Structure | |||||||||
| Mechanism of Pergolide-Amisulpride Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
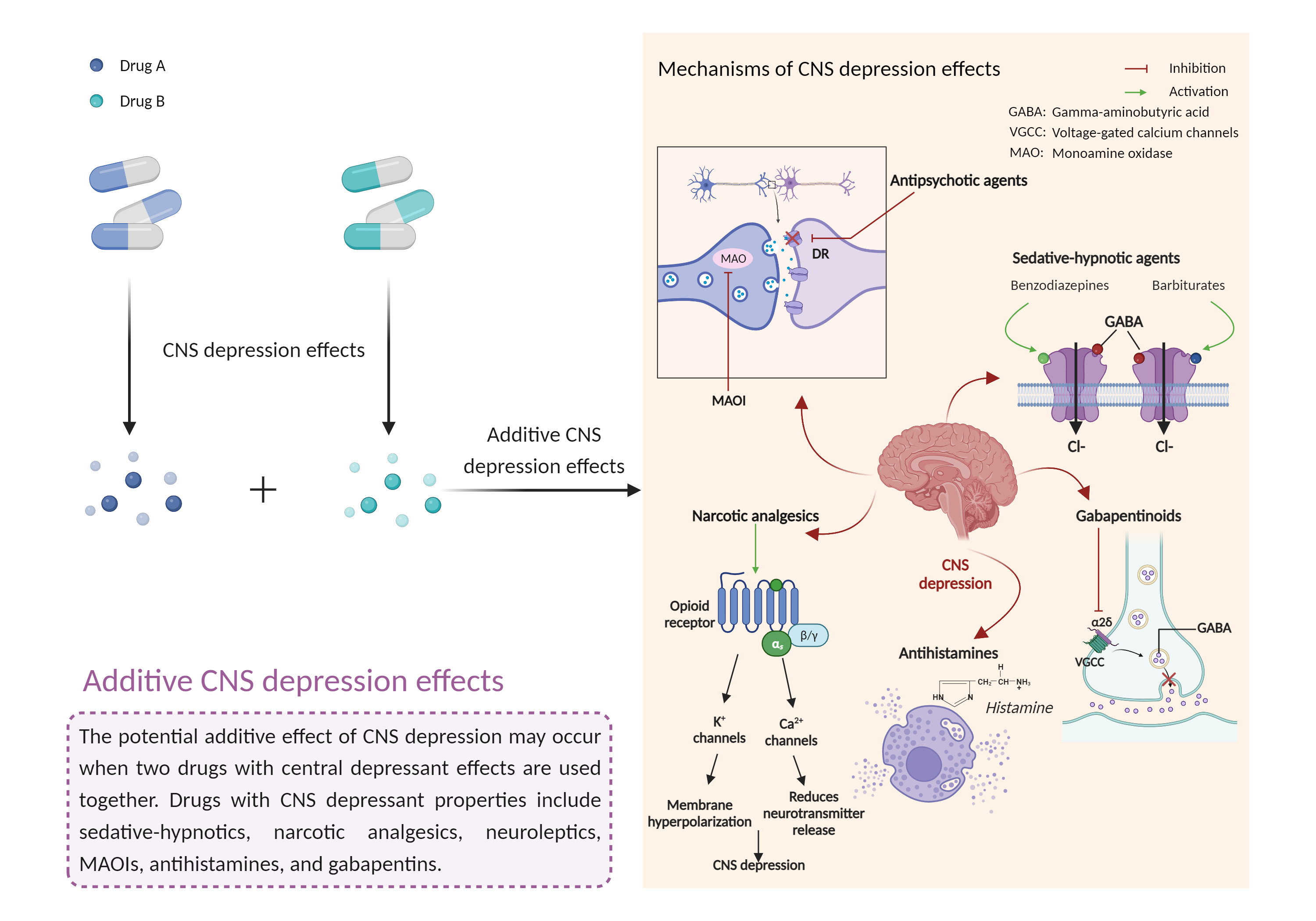
| Additive CNS depression effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Pergolide | Amisulpride | |||||||
| Mechanism 1 | CNS depression effects | CNS depression effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | CNS depression effects | ||||||||
| Factor Description | CNS depressants are drugs that inhibit or suppress brain activity and can reduce mental and physical processes. Excessive CNS depression can lead to decreased heart rate, slow breathing (less than 10 breaths per minute), extreme confusion or loss of memory, nausea and vomiting, poor judgment, blue lips or fingertips, irritability and aggression, and clammy or cold skin. | ||||||||
| Mechanism Description |
|
||||||||
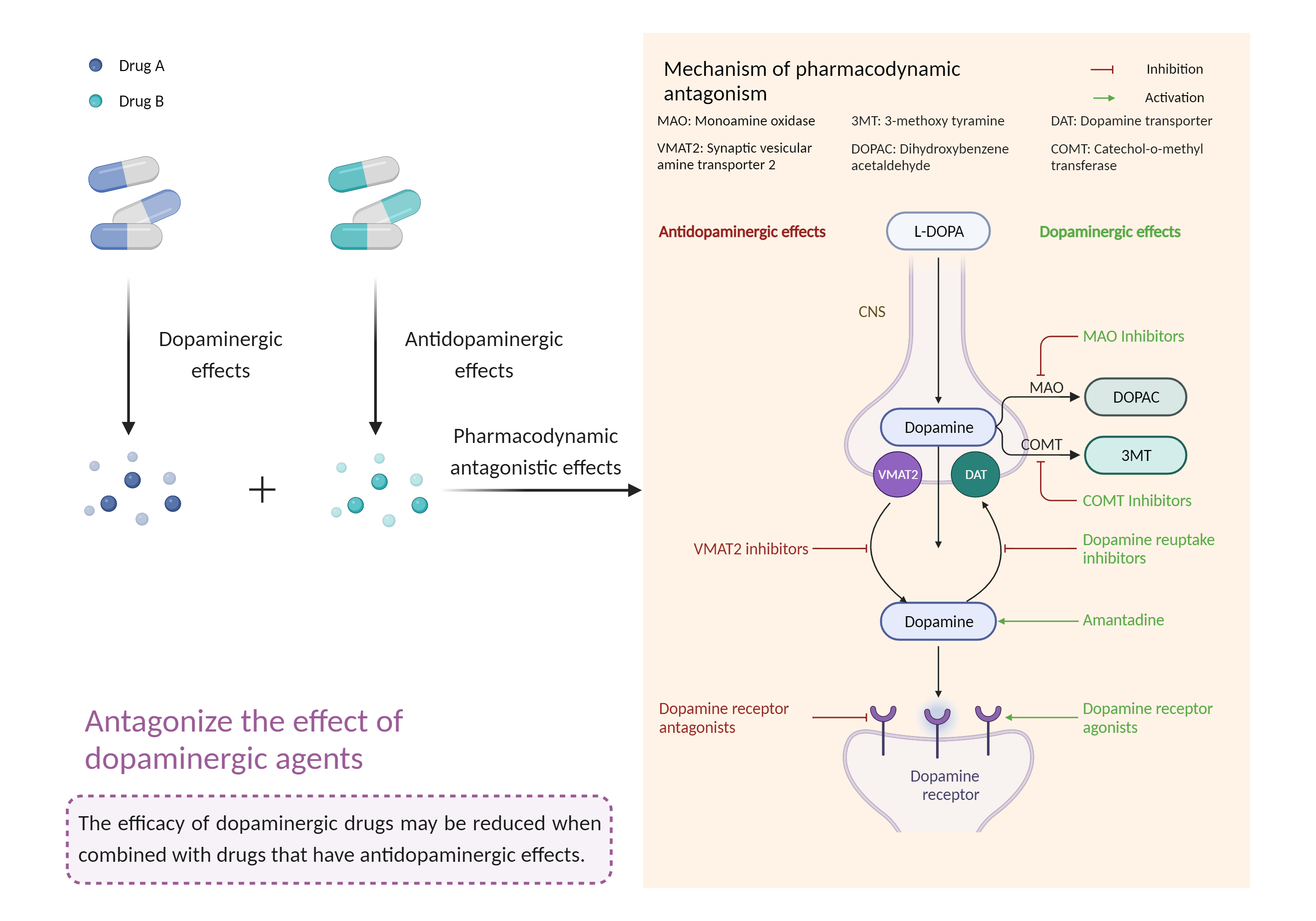
| Antagonize the effect of dopaminergic agents Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Pergolide | Amisulpride | |||||||
| Mechanism 2 |
Dopaminergic agent Dopamine receptor Agonist |
Antidopaminergic effects Dopamine receptor Antagonist |
|||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Dopamine receptor | Structure Sequence | |||||||
| Protein Family | G-protein coupled receptor 1 family | ||||||||
| Protein Function |
Dopamine receptor whose activity is mediated by G proteins which activate adenylyl cyclase.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Coadministration of amisulpride with levodopa or other dopamine agonists should generally be avoided. If concomitant administration is necessary, patients should be monitored for excessive sedation, hypotension, and potentially diminished therapeutic response to both treatments. | ||||||||
