Details of Drug-Drug Interaction
| Drug General Information (ID: DDI1AH7Z8B) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Gemfibrozil | Drug Info | Selexipag | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihyperammonia Agents | Agents For Pulmonary Hypertension | |||||||
| Structure | |||||||||
| Mechanism of Gemfibrozil-Selexipag Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
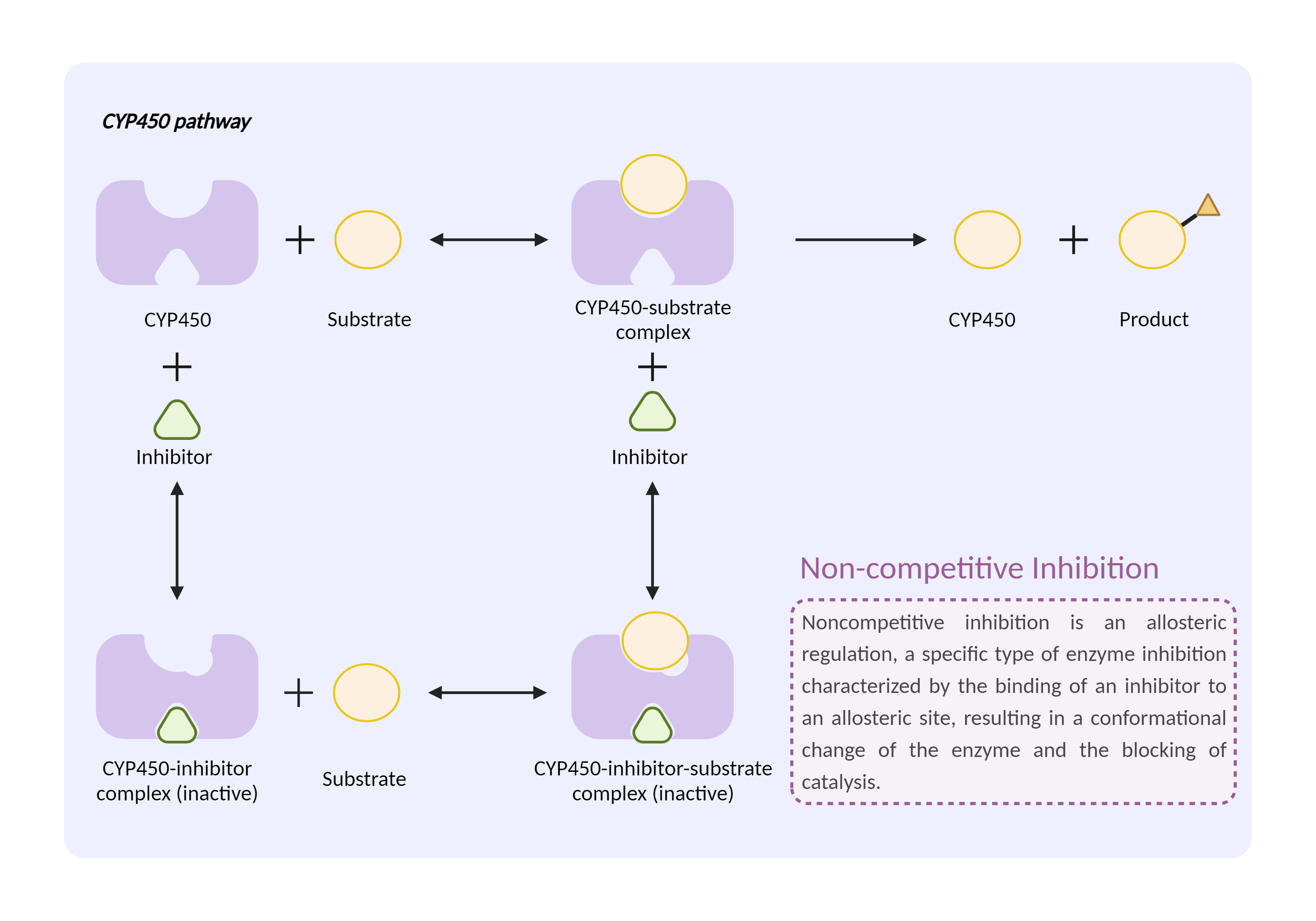
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Gemfibrozil | Selexipag | |||||||
| Mechanism | CYP450 2C8 inhibitor | CYP450 2C8 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2C8 |
×
Structure
Sequence
MEPFVVLVLCLSFMLLFSLWRQSCRRRKLPPGPTPLPIIGNMLQIDVKDICKSFTNFSKVYGPVFTVYFGMNPIVVFHGYEAVKEALIDNGEEFSGRGNSPISQRITKGLGIISSNGKRWKEIRRFSLTTLRNFGMGKRSIEDRVQEEAHCLVEELRKTKASPCDPTFILGCAPCNVICSVVFQKRFDYKDQNFLTLMKRFNENFRILNSPWIQVCNNFPLLIDCFPGTHNKVLKNVALTRSYIREKVKEHQASLDVNNPRDFIDCFLIKMEQEKDNQKSEFNIENLVGTVADLFVAGTETTSTTLRYGLLLLLKHPEVTAKVQEEIDHVIGRHRSPCMQDRSHMPYTDAVVHEIQRYSDLVPTGVPHAVTTDTKFRNYLIPKGTTIMALLTSVLHDDKEFPNPNIFDPGHFLDKNGNFKKSDYFMPFSAGKRICAGEGLARMELFLFLTTILQNFNLKSVDDLKNLNTTAVTKGIVSLPPSYQICFIPV
|
|||||||
| Gene Name | CYP2C8 | ||||||||
| Uniprot ID | CP2C8_HUMAN | ||||||||
| KEGG Pathway | hsa:1558 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of various endogenous substrates, including fatty acids, steroid hormones and vitamins (PubMed:7574697, PubMed:11093772, PubMed:14559847, PubMed:15766564, PubMed:19965576). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:7574697, PubMed:11093772, PubMed:14559847, PubMed:15766564, PubMed:19965576). Primarily catalyzes the epoxidation of double bonds of polyunsaturated fatty acids (PUFA) with a preference for the last double bond (PubMed:7574697, PubMed:15766564, PubMed:19965576). Catalyzes the hydroxylation of carbon-hydrogen bonds. Metabolizes all trans-retinoic acid toward its 4-hydroxylated form (PubMed:11093772). Displays 16-alpha hydroxylase activity toward estrogen steroid hormones, 17beta-estradiol (E2) and estrone (E1) (PubMed:14559847). Plays a role in the oxidative metabolism of xenobiotics. It is the principal enzyme responsible for the metabolism of the anti-cancer drug paclitaxel (taxol) (PubMed:26427316).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of selexipag with potent CYP450 2C8 inhibitors is contraindicated by the manufacturer. | ||||||||
