Details of Drug-Drug Interaction
| Drug General Information (ID: DDI0SEDUK7) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Omacetaxine mepesuccinate | Drug Info | Siltuximab | Drug Info | |||||
| Drug Type | Small molecule | Monoclonal antibody | |||||||
| Therapeutic Class | Antineoplastics | Interleukin Inhibitors | |||||||
| Mechanism of Omacetaxine mepesuccinate-Siltuximab Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive immunosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
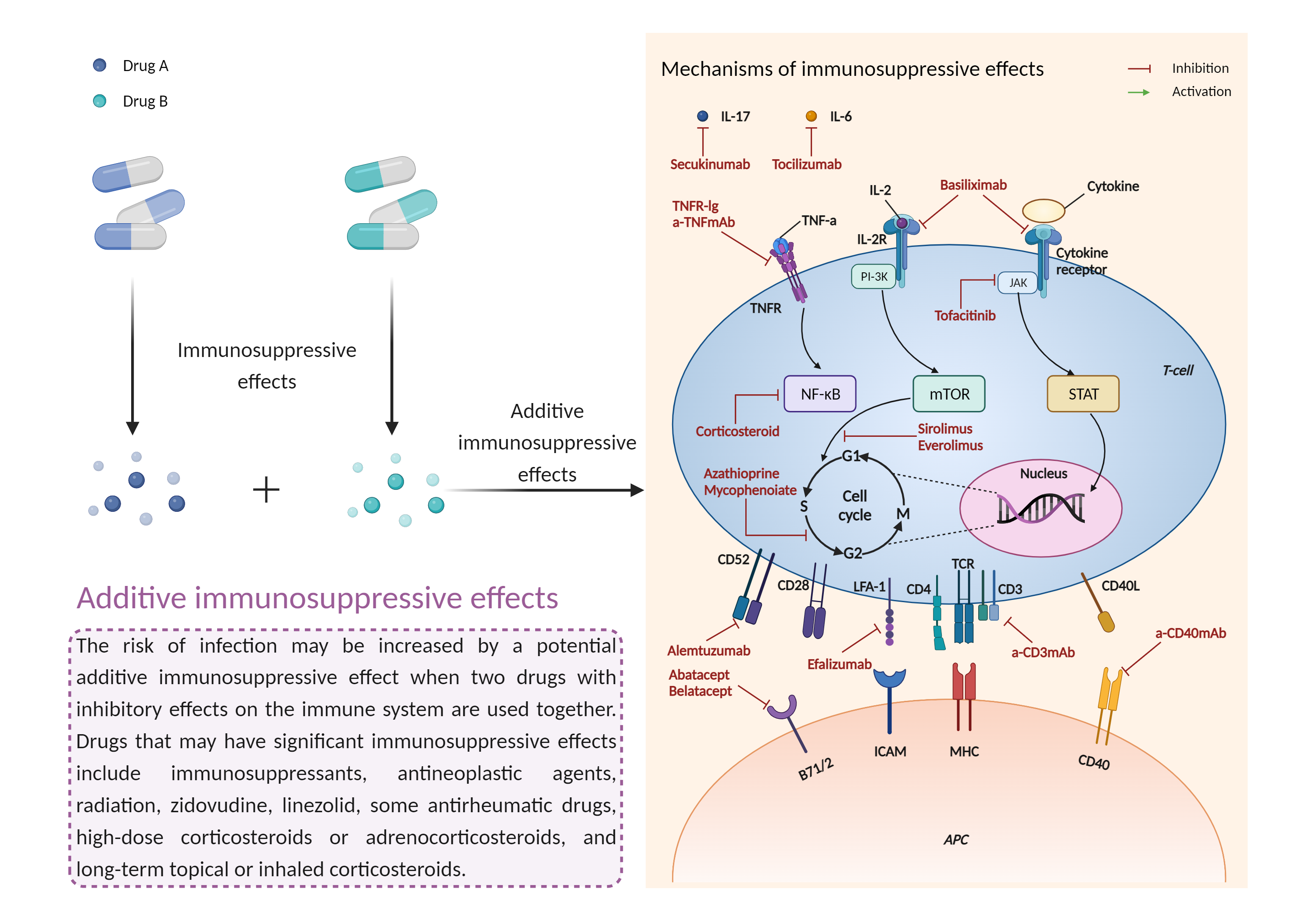 |
|||||||||
| Drug Name | Omacetaxine mepesuccinate | Siltuximab | |||||||
| Mechanism | Immunosuppressive effects | Immunosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Immunosuppressive effects | ||||||||
| Factor Description | Immunosuppression is when your immune system is not functioning as it should. The immune system is made up of cells, tissues and organs that help the body fight off infections. If the immune system is suppressed, an infection that your body was able to control may become serious or even fatal. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if omacetaxine must be used in patients who have recently received or are receiving treatment with other immunosuppressive or myelosuppressive drugs, and vice versa. Close clinical and laboratory monitoring for the development of severe hematologic adverse effects is recommended both during and after discontinuation of therapy. Patients should be advised to seek medical attention if they experience symptoms such as fever, chills, shortness of breath, fatigue, and any unusual bleeding or bruising. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Synribo (omacetaxine). Teva Pharmaceuticals USA, North Wales, PA. | ||||||||||||||||||
