Details of Drug-Drug Interaction
| Drug General Information (ID: DDI0DNX4T5) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Iohexol | Drug Info | Urea | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Non-Ionic Iodinated Contrast Media | Topical Emollients | |||||||
| Structure | |||||||||
| Mechanism of Iohexol-Urea Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
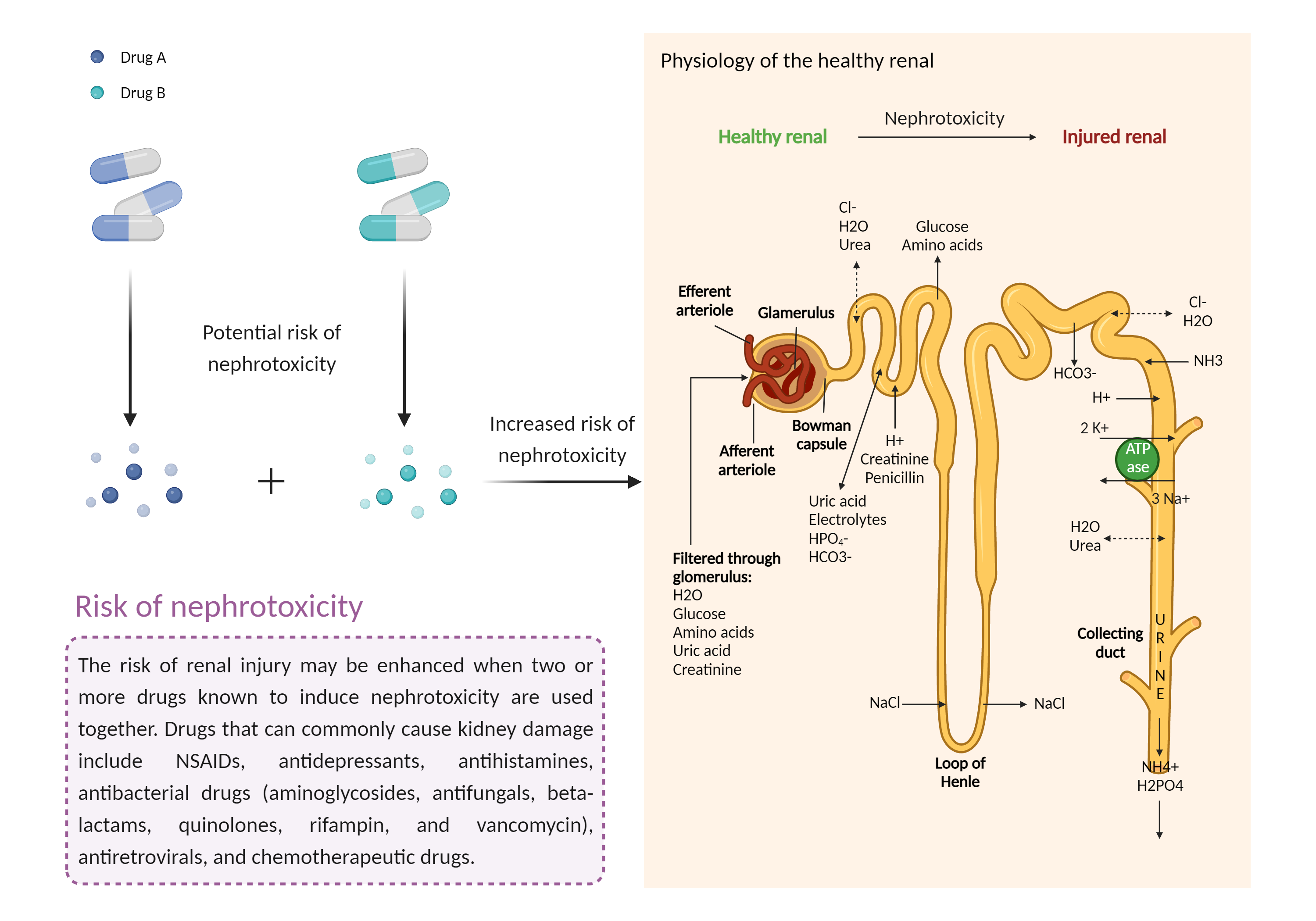
| Increased risk of nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Iohexol | Urea | |||||||
| Mechanism 1 | Nephrotoxicity | Nephrotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Mechanism 2 | Nephrotoxicity |
Nephrotoxicity Decrease renal perfusion |
|||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Mechanism 3 | Nephrotoxicity | Decrease renal perfusion/blood flow rate | |||||||
| Key Mechanism Factor 3 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Whenever possible, alternative imaging techniques should be considered in patients who are at high risk for contrast-induced nephropathy. Otherwise, some experts recommend discontinuing diuretics 1 to 2 days before administration of contrast media, depending on the clinical feasibility of doing so. The smallest effective dose of a nonionic, hypo- or iso-osmolar contrast medium (e.g., iohexol, iodixanol, iopamidol) should be used, since the risk of nephropathy is increased with increasing contrast dose and/or osmolarity. Repeat procedures with contrast media, if necessary, should not occur until at least 72 hours after the previous contrast exposure and renal function has fully recovered. Although it is not necessary to measure the serum creatinine levels of every patient before contrast administration, measurements should generally be made in patients receiving contrast agent by intraarterial administration (which is associated with increased risk of nephropathy relative to intravenous administration) and patients with a history of kidney disease, proteinuria, kidney surgery, diabetes, hypertension, gout, or other risk factors for nephropathy. Creatinine measurements should be continued for 24 to 48 hours after administration of contrast medium. It is important that patients be adequately hydrated with either saline or sodium bicarbonate. | ||||||||
