Details of Drug-Drug Interaction
| Drug General Information (ID: DDIZYJ2FDQ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Tizanidine | Drug Info | Cyclandelate | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Analgesics | Vasodilator Agents | |||||||
| Structure | |||||||||
| Mechanism of Tizanidine-Cyclandelate Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
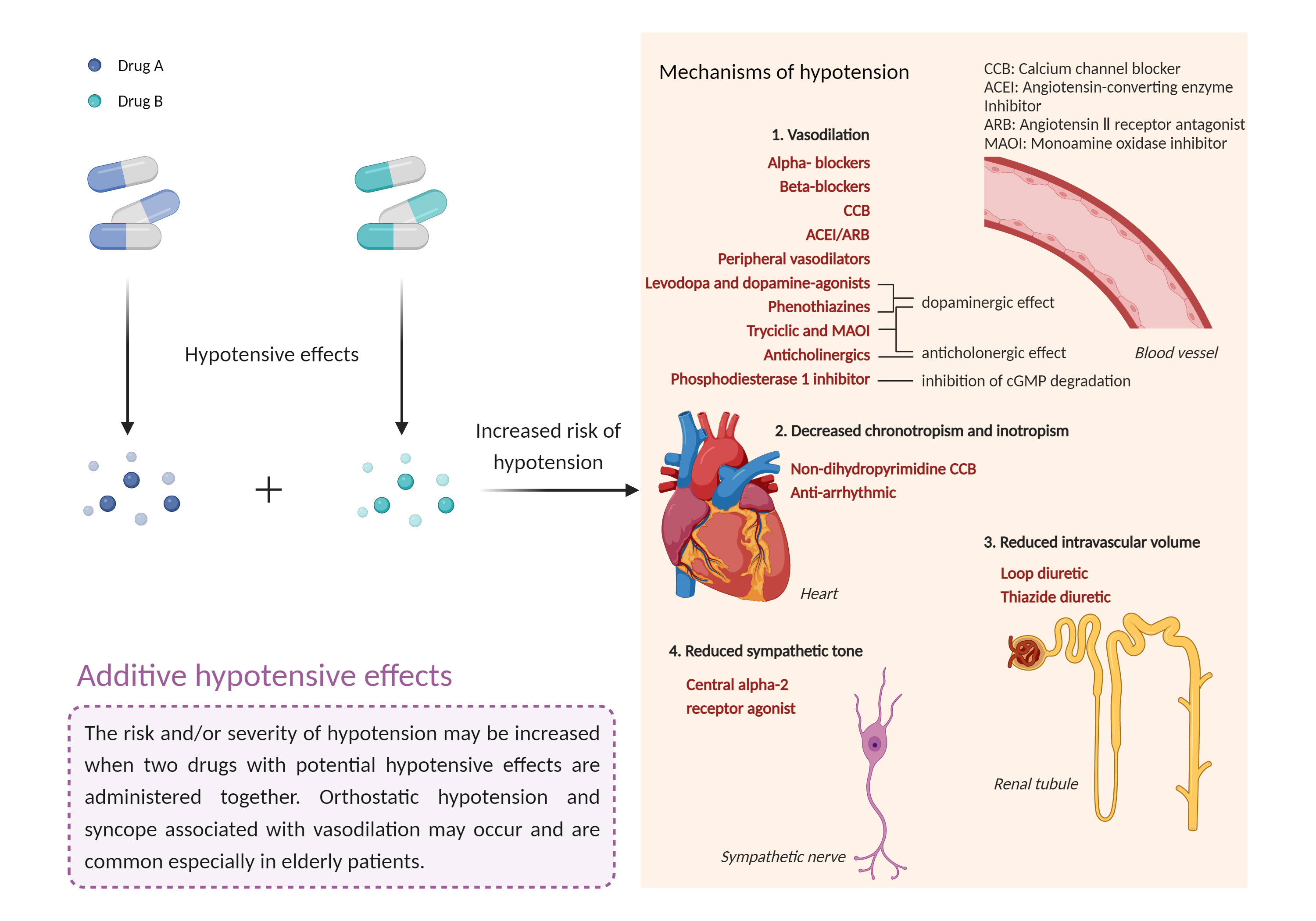
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Tizanidine | Cyclandelate | |||||||
| Mechanism |
Hypotensive effects Alpha-2 adrenergic receptor Agonist |
Hypotensive effects Voltage-gated calcium channel Blocker |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Adrenergic receptor alpha-2 | Structure Sequence | |||||||
| Protein Family | G-protein coupled receptor 1 family | ||||||||
| Protein Function |
Alpha-2 adrenergic receptors mediate the catecholamine-induced inhibition of adenylate cyclase through the action of G proteins. The rank order of potency for agonists of this receptor is oxymetazoline > clonidine > epinephrine > norepinephrine > phenylephrine > dopamine > p-synephrine > p-tyramine > serotonin = p-octopamine. For antagonists, the rank order is yohimbine > phentolamine = mianserine > chlorpromazine = spiperone = prazosin > propanolol > alprenolol = pindolol.
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Voltage-dependent L-type calcium channel | Structure Sequence | |||||||
| Protein Family | Calcium channel beta subunit family | ||||||||
| Protein Function |
Regulatory subunit of L-type calcium channels (PubMed:1309651, PubMed:8107964, PubMed:15615847). Regulates the activity of L-type calcium channels that contain CACNA1A as pore-forming subunit (By similarity). Regulates the activity of L-type calcium channels that contain CACNA1C as pore-forming subunit and increases the presence of the channel complex at the cell membrane (PubMed:15615847). Required for functional expression L-type calcium channels that contain CACNA1D as pore-forming subunit (PubMed:1309651). Regulates the activity of L-type calcium channels that contain CACNA1B as pore-forming subunit (PubMed:8107964).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | A lower initial dosage and cautious dosage titration should be considered when tizanidine is initiated in patients receiving hypotensive medications. Although single doses of less than 8 mg of tizanidine have not been shown effective for spasticity in controlled clinical studies, it may be prudent to initiate treatment with 4 mg doses and gradually increase in 2 to 4 mg increments until optimum effect is achieved. The dose can be repeated at 6 to 8 hour intervals as needed, up to a maximum of three doses in 24 hours and a total daily dosage of 36 mg. However, experience with single doses exceeding 8 mg and daily doses exceeding 24 mg is limited. Close monitoring for development of hypotension is recommended. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Zanaflex (tizanidine). Acorda Therapeutics, Hawthorne, NY. | ||||||||||||||||||
