Details of Drug-Drug Interaction
| Drug General Information (ID: DDIZO5VWRH) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Zalcitabine | Drug Info | Tafamidis | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anti-Hiv Agents | Transthyretin Stabilizers | |||||||
| Structure | |||||||||
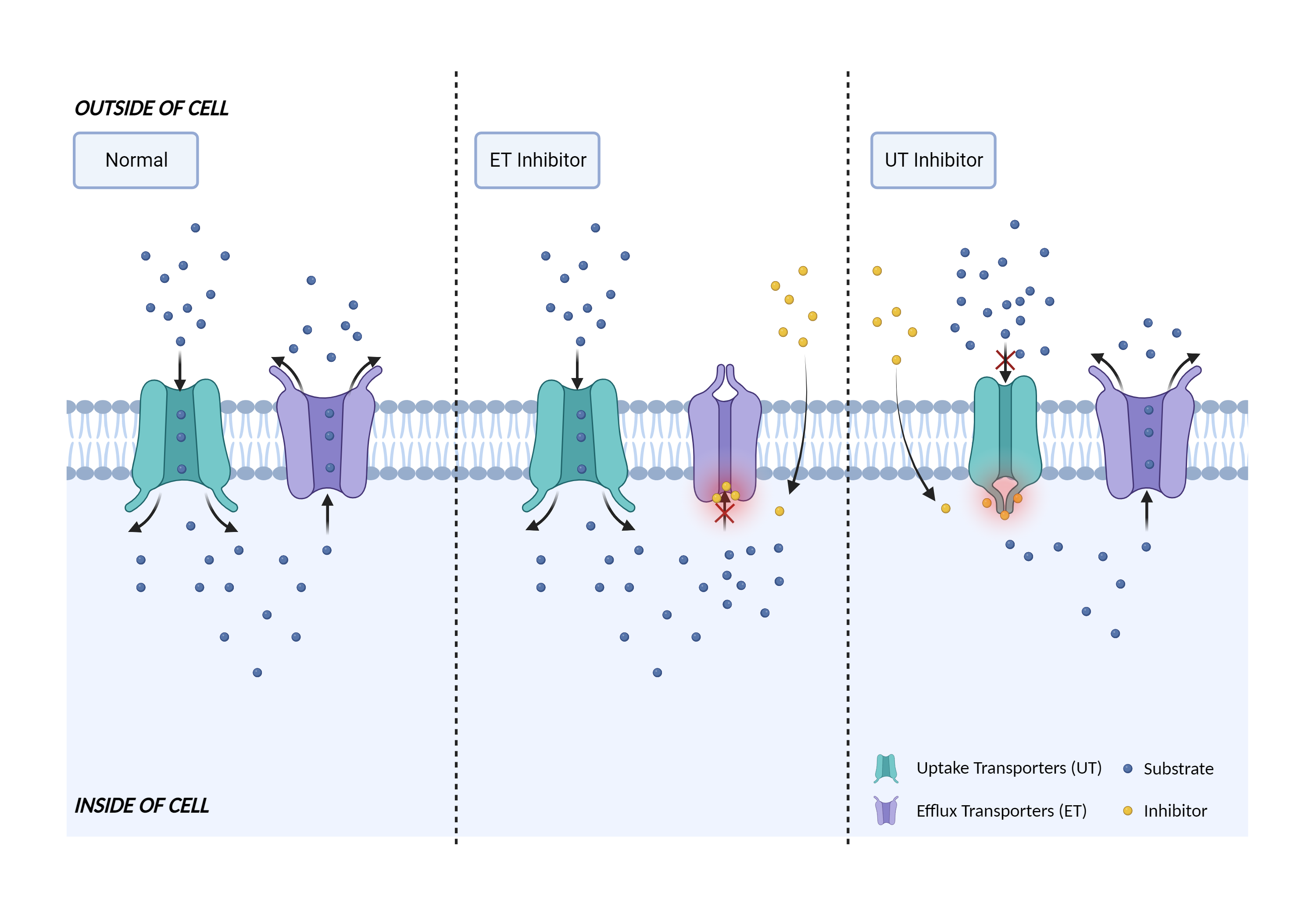
| Mechanism of Zalcitabine-Tafamidis Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Transporter inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Zalcitabine | Tafamidis | |||||||
| Mechanism 1 | OAT1 substrate | OAT1 inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Solute carrier family 22 member 6 |
×
Structure
Sequence
MAFNDLLQQVGGVGRFQQIQVTLVVLPLLLMASHNTLQNFTAAIPTHHCRPPADANLSKNGGLEVWLPRDRQGQPESCLRFTSPQWGLPFLNGTEANGTGATEPCTDGWIYDNSTFPSTIVTEWDLVCSHRALRQLAQSLYMVGVLLGAMVFGYLADRLGRRKVLILNYLQTAVSGTCAAFAPNFPIYCAFRLLSGMALAGISLNCMTLNVEWMPIHTRACVGTLIGYVYSLGQFLLAGVAYAVPHWRHLQLLVSAPFFAFFIYSWFFIESARWHSSSGRLDLTLRALQRVARINGKREEGAKLSMEVLRASLQKELTMGKGQASAMELLRCPTLRHLFLCLSMLWFATSFAYYGLVMDLQGFGVSIYLIQVIFGAVDLPAKLVGFLVINSLGRRPAQMAALLLAGICILLNGVIPQDQSIVRTSLAVLGKGCLAASFNCIFLYTGELYPTMIRQTGMGMGSTMARVGSIVSPLVSMTAELYPSMPLFIYGAVPVAASAVTVLLPETLGQPLPDTVQDLESRWAPTQKEAGIYPRKGKQTRQQQEHQKYMVPLQASAQEKNGL
|
|||||||
| Gene Name | OAT1 | ||||||||
| Uniprot ID | S22A6_HUMAN | ||||||||
| KEGG Pathway | hsa:9356 | ||||||||
| Protein Family | Major facilitator (TC 2.A.1) superfamily | ||||||||
| Protein Function |
Involved in the renal elimination of endogenous and exogenous organic anions. Functions as organic anion exchanger when the uptake of one molecule of organic anion is coupled with an efflux of one molecule of endogenous dicarboxylic acid (glutarate, ketoglutarate, etc). Mediates the sodium-independent uptake of 2,3-dimercapto-1-propanesulfonic acid (DMPS) (By similarity). Mediates the sodium-independent uptake of p-aminohippurate (PAH), ochratoxin (OTA), acyclovir (ACV), 3'-azido-3-'deoxythymidine (AZT), cimetidine (CMD), 2,4-dichloro-phenoxyacetate (2,4-D), hippurate (HA), indoleacetate (IA), indoxyl sulfate (IS) and 3-carboxy-4-methyl-5-propyl-2-furanpropionate (CMPF), cidofovir, adefovir, 9-(2-phosphonylmethoxyethyl) guanine (PMEG), 9-(2-phosphonylmethoxyethyl) diaminopurine (PMEDAP) and edaravone sulfate. PAH uptake is inhibited by p-chloromercuribenzenesulphonate (PCMBS), diethyl pyrocarbonate (DEPC), sulindac, diclofenac, carprofen, glutarate and okadaic acid (By similarity). PAH uptake is inhibited by benzothiazolylcysteine (BTC), S-chlorotrifluoroethylcysteine (CTFC), cysteine S-conjugates S-dichlorovinylcysteine (DCVC), furosemide, steviol, phorbol 12-myristate 13-acetate (PMA), calcium ionophore A23187, benzylpenicillin, furosemide, indomethacin, bumetamide, losartan, probenecid, phenol red, urate, and alpha-ketoglutarate.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Mechanism 2 | OAT3 substrate | OAT3 inhibitor | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Solute carrier family 22 member 8 |
×
Structure
Sequence
MTFSEILDRVGSMGHFQFLHVAILGLPILNMANHNLLQIFTAATPVHHCRPPHNASTGPWVLPMGPNGKPERCLRFVHPPNASLPNDTQRAMEPCLDGWVYNSTKDSIVTEWDLVCNSNKLKEMAQSIFMAGILIGGLVLGDLSDRFGRRPILTCSYLLLAASGSGAAFSPTFPIYMVFRFLCGFGISGITLSTVILNVEWVPTRMRAIMSTALGYCYTFGQFILPGLAYAIPQWRWLQLTVSIPFFVFFLSSWWTPESIRWLVLSGKSSKALKILRRVAVFNGKKEEGERLSLEELKLNLQKEISLAKAKYTASDLFRIPMLRRMTFCLSLAWFATGFAYYSLAMGVEEFGVNLYILQIIFGGVDVPAKFITILSLSYLGRHTTQAAALLLAGGAILALTFVPLDLQTVRTVLAVFGKGCLSSSFSCLFLYTSELYPTVIRQTGMGVSNLWTRVGSMVSPLVKITGEVQPFIPNIIYGITALLGGSAALFLPETLNQPLPETIEDLENWSLRAKKPKQEPEVEKASQRIPLQPHGPGLGSS
|
|||||||
| Gene Name | OAT3 | ||||||||
| Uniprot ID | S22A8_HUMAN | ||||||||
| KEGG Pathway | hsa:9376 | ||||||||
| Protein Family | Major facilitator (TC 2.A.1) superfamily | ||||||||
| Protein Function |
Plays an important role in the excretion/detoxification of endogenous and exogenous organic anions, especially from the brain and kidney. Involved in the transport basolateral of steviol, fexofenadine. Transports benzylpenicillin (PCG), estrone-3-sulfate (E1S), cimetidine (CMD), 2,4-dichloro-phenoxyacetate (2,4-D), p-amino-hippurate (PAH), acyclovir (ACV) and ochratoxin (OTA).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if tafamidis is used concomitantly with drugs that are substrates of these transport proteins, particularly those with a narrow therapeutic range. Dosage adjustments as well as clinical and laboratory monitoring should be considered whenever tafamidis is added to or withdrawn from therapy with these drugs. Patients should be monitored for the development of adverse effects. | ||||||||
