Details of Drug-Drug Interaction
| Drug General Information (ID: DDIYTDSWVJ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Eplerenone | Drug Info | Eprosartan | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Antihypertensive Agents | |||||||
| Structure | |||||||||
| Mechanism of Eplerenone-Eprosartan Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
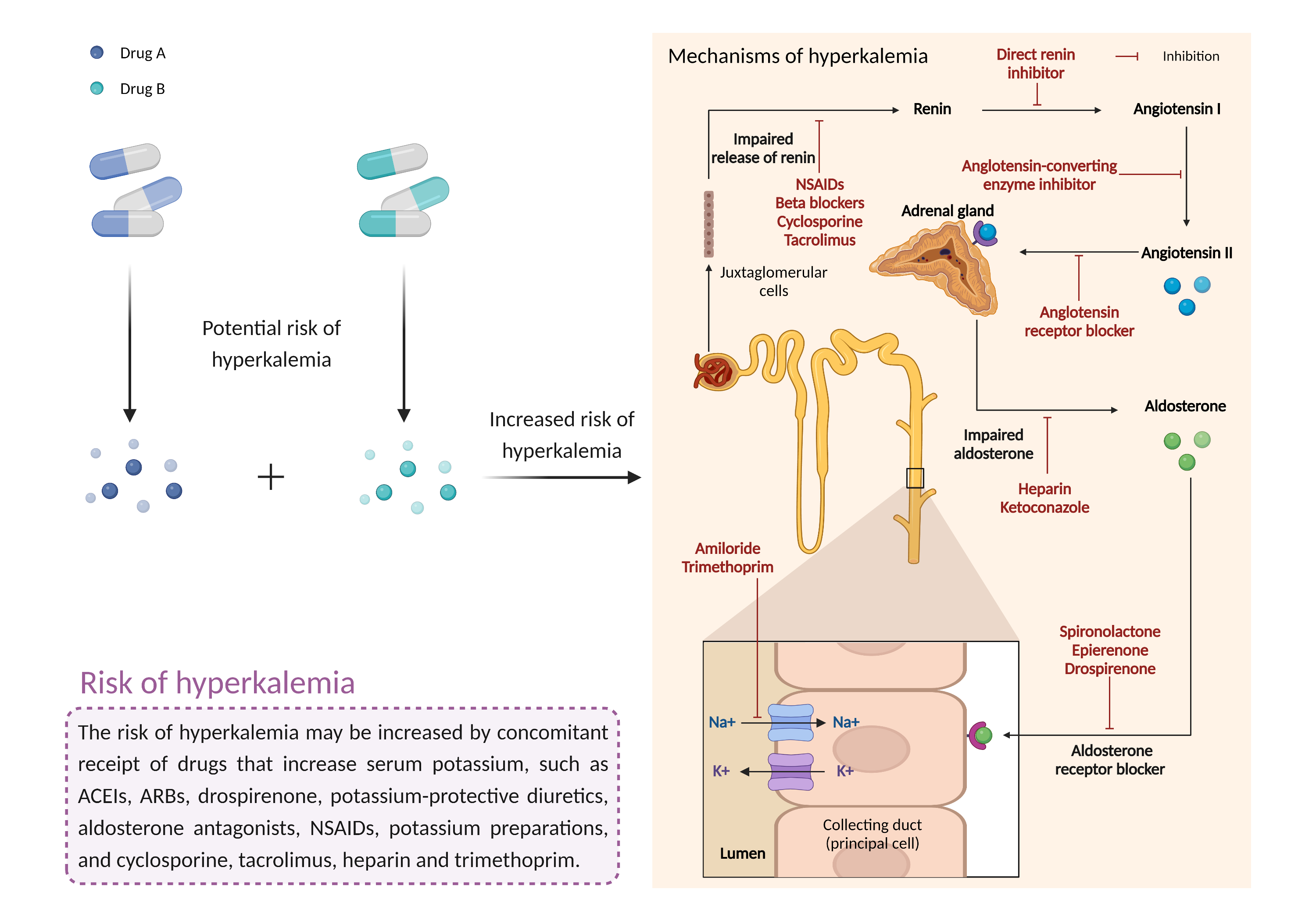
| Increased risk of hyperkalemia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Eplerenone | Eprosartan | |||||||
| Mechanism | Hyperkalemia | Hyperkalemia | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hyperkalemia | ||||||||
| Factor Description | Hyperkalemia is a condition in which the level of potassium in the blood is higher than normal. While mild cases may not produce symptoms, severe hyperkalemia can lead to fatal arrhythmias if left untreated. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Given the potential for serious, sometimes fatal cardiac arrhythmias associated with hyperkalemia, caution is advised if eplerenone is administered with ACE inhibitors or angiotensin II receptor antagonists. Periodic monitoring of serum potassium levels is recommended until the effect of eplerenone is established. In clinical trials, monitoring occurred every 2 weeks for the first 1 to 2 months, then monthly thereafter. Particular caution is warranted in patients with renal impairment, diabetes, old age, or dehydration. Patients should be advised to seek medical attention if they experience signs and symptoms of hyperkalemia such as nausea, vomiting, weakness, listlessness, tingling of the extremities, paralysis, confusion, weak pulse, and a slow or irregular heartbeat. | ||||||||
