Details of Drug-Drug Interaction
| Drug General Information (ID: DDIYP621U5) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Guanethidine | Drug Info | Iobenguane (I-131) | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Therapeutic Radiopharmaceuticals | |||||||
| Structure | |||||||||
| Mechanism of Guanethidine-Iobenguane (I-131) Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
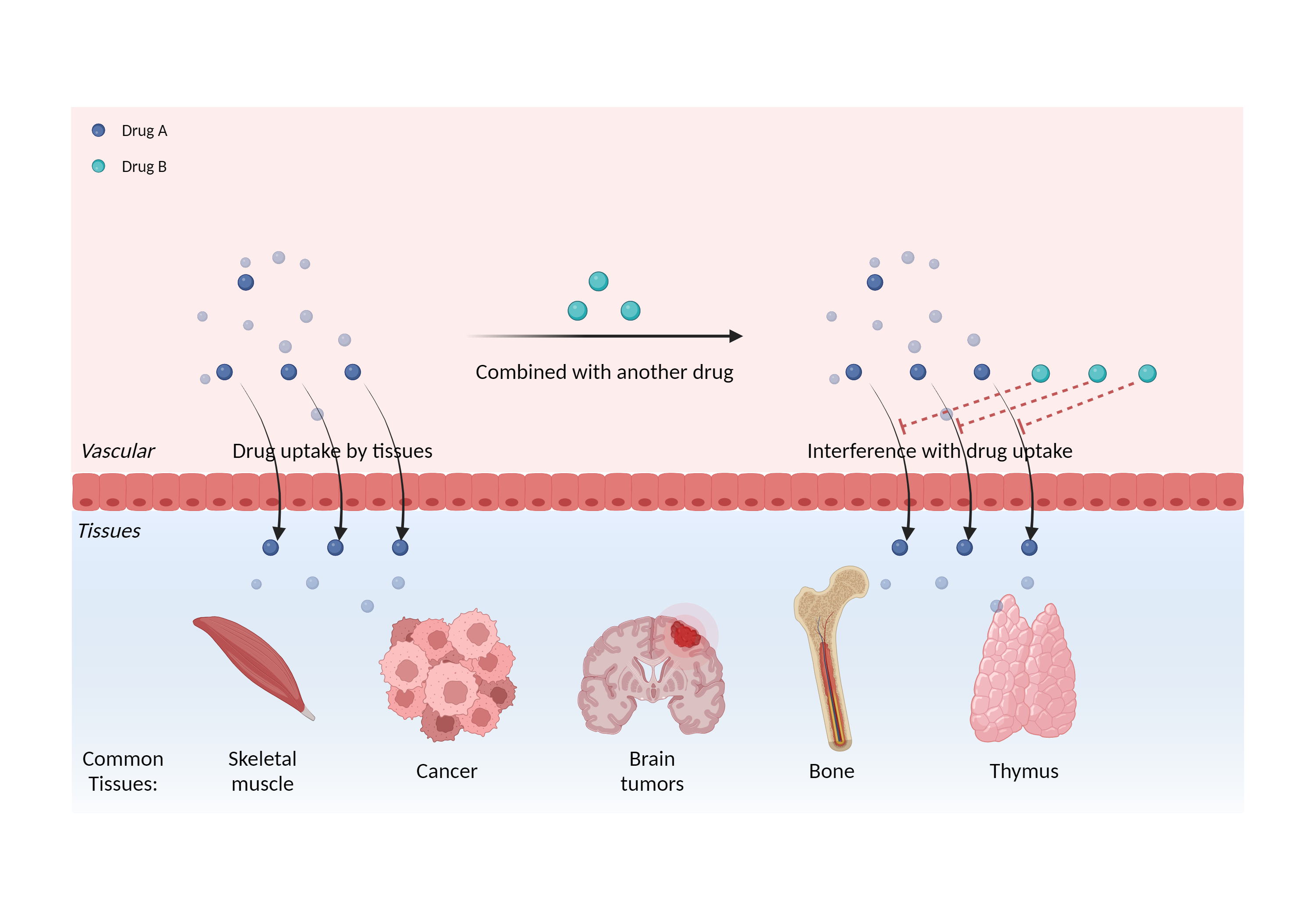
| Interference of cell/tissue uptake Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Guanethidine | Iobenguane (I-131) | |||||||
| Mechanism | Interference with radiolabeled norepinephrine analog effects | Radiolabeled norepinephrine analog | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Pharmacokinetics | ||||||||
| Factor Description | Alters the distribution of the drug, leading to changes in the plasma concentration of the drug and affecting the pharmacological effects. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Drugs that reduce catecholamine uptake or deplete catecholamine stores should be discontinued for at least five biological half-lives before administration of either the dosimetry dose or a therapeutic dose of iobenguane I-131. Do not administer these drugs until at least 7 days after each iobenguane I-131 dose. Patients should be monitored for the occurrence of clinically significant withdrawal symptoms, especially patients with elevated levels of circulating catecholamines and their metabolites. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Azedra (iobenguane I-131). Progenics Pharmaceuticals, Inc., New York, NY. | ||||||||||||||||||
