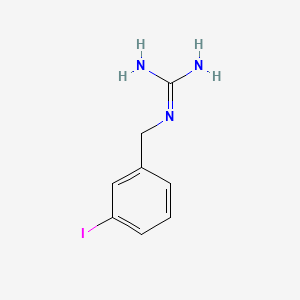
Details of Drug
| Drug-Drug Interaction Network | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sunburst Graph |
AM003 Affected organization distribution
AM005 Affected excretion pathways
AM006 Pharmacodynamic additive effects
AM007 Pharmacodynamic antagonistic effects
BM014 Interference of cell/tissue uptake
BM027 Decreased renal excretion due to nephrotoxicity
BM043 Additive immunosuppressive effects
BM044 Additive myelosuppressive effects
BM072 Increased risk of nephrotoxicity
BM100 Antagonize the effect of vaccine/toxoid
BM105 Antagonize the effect of probiotics
BM106 Diminish the response to diagnostic skin test antigens
BM107 Attenuated pharmacological effects (Unspecific)
BM109 Increased risk of other adverse reactions (Unspecific)
|
||||||||
| Relation Graph | |||||||||
| Full list of drugs interacting with Iobenguane (I-131) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Affected organization distribution | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Interference of cell/tissue uptake | Drug Num: 81 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0068
|
Amitriptyline
|
C20H23N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0072
|
Amoxapine
|
C17H16ClN3O
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0074
|
Amphetamine
|
C9H13N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0165
|
Benzphetamine
|
C17H21N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0200
|
Bretylium
|
C11H17BrN+
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0220
|
Bupropion
|
C13H18ClNO
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0265
|
Carteolol
|
C16H24N2O3
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0266
|
Carteolol (ophthalmic)
|
C16H24N2O3
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0267
|
Carvedilol
|
C24H26N2O4
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0361
|
Clomipramine
|
C19H23ClN2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0378
|
Cocaine (topical)
|
C17H21NO4
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0444
|
Deserpidine
|
C32H38N2O8
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0446
|
Desipramine
|
C18H22N2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0451
|
Desvenlafaxine
|
C16H25NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0457
|
Dexfenfluramine
|
C12H16F3N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0465
|
Dextroamphetamine
|
C9H13N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0486
|
Diethylpropion
|
C13H19NO
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0517
|
Dopamine
|
C8H11NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0523
|
Doxapram
|
C24H30N2O2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0525
|
Doxepin
|
C19H21NO
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0526
|
Doxepin (topical)
|
C19H21NO
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0539
|
Duloxetine
|
C18H19NOS
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0579
|
Ephedrine
|
C10H15NO
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0580
|
Ephedrine (nasal)
|
C10H15NO
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0593
|
Ergotamine
|
C33H35N5O5
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0641
|
Fenfluramine
|
C12H16F3N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0743
|
Guanadrel
|
C10H19N3O2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0744
|
Guanethidine
|
C10H22N4
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0795
|
Imipramine
|
C19H24N2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0920
|
Levomilnacipran
|
C15H22N2O
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1036
|
Methylene blue
|
C16H18ClN3S
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1107
|
Naphazoline (nasal)
|
C14H14N2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1108
|
Naphazoline (ophthalmic)
|
C14H14N2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1211
|
Ozanimod
|
C23H24N4O3
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1358
|
Propylhexedrine (nasal)
|
C10H21N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1567
|
Tetryzoline (nasal)
|
C13H16N2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1568
|
Tetryzoline (ophthalmic)
|
C13H16N2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0853
|
Isocarboxazid
|
C12H13N3O2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0884
|
Labetalol
|
C19H24N2O3
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0933
|
Linezolid
|
C16H20FN3O4
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0937
|
Lisdexamfetamine
|
C15H25N3O
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0979
|
Maprotiline
|
C20H23N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0981
|
Mazindol
|
C16H13ClN2O
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1002
|
Mephentermine
|
C11H17N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1012
|
Metamfetamine
|
C10H15N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1013
|
Metaraminol
|
C9H13NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1039
|
Methylphenidate
|
C14H19NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1061
|
Midodrine
|
C12H18N2O4
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1066
|
Milnacipran
|
C15H22N2O
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1158
|
Nortriptyline
|
C19H21N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1204
|
Oxymetazoline (nasal)
|
C16H24N2O
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1205
|
Oxymetazoline (ophthalmic)
|
C16H24N2O
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1244
|
Penbutolol
|
C18H29NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1262
|
Phendimetrazine
|
C12H17NO
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1263
|
Phenelzine
|
C8H12N2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1270
|
Phentermine
|
C10H15N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1276
|
Phenylephrine
|
C9H13NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1277
|
Phenylephrine (nasal)
|
C9H13NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1278
|
Phenylephrine (ophthalmic)
|
C9H13NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1280
|
Phenylephrine (topical)
|
C9H13NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1281
|
Phenylpropanolamine
|
C9H13NO
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1293
|
Pindolol
|
C14H20N2O2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1345
|
Procarbazine
|
C12H19N3O
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1357
|
Propranolol
|
C16H21NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1361
|
Protriptyline
|
C19H21N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1363
|
Pseudoephedrine
|
C10H15NO
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1385
|
Rasagiline
|
C12H13N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1393
|
Reserpine
|
C33H40N2O9
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1437
|
Safinamide
|
C17H19FN2O2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1455
|
Selegiline
|
C13H17N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1498
|
Sotalol
|
C12H20N2O3S
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1502
|
St. John's Wort
|
NA
|
NA
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1535
|
Tapentadol
|
C14H23NO
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1585
|
Timolol
|
C13H24N4O3S
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1586
|
Timolol (ophthalmic)
|
C13H24N4O3S
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1616
|
Tramadol
|
C16H25NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1620
|
Tranylcypromine
|
C9H11N
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1647
|
Trimipramine
|
C20H26N2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1689
|
Venlafaxine
|
C17H27NO2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1712
|
Xylometazoline (nasal)
|
C16H24N2
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1713
|
Yohimbine
|
C21H26N2O3
|
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Affected excretion pathways | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Decreased renal excretion due to nephrotoxicity | Drug Num: 6 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0576
|
Entecavir
|
C12H15N5O3
|
|
Moderate | Inter Info |
[51]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0668
|
Flucytosine
|
C4H4FN3O
|
|
Moderate | Inter Info |
[55]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1389
|
Remdesivir
|
C27H35N6O8P
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1193
|
Oxaliplatin
|
C8H14N2O4Pt
|
|
Moderate | Inter Info |
[80]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1241
|
Pemetrexed
|
C20H21N5O6
|
|
Moderate | Inter Info |
[84]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1543
|
Telbivudine
|
C10H14N2O5
|
|
Moderate | Inter Info |
[86]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pharmacodynamic additive effects | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Additive immunosuppressive effects | Drug Num: 39 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0023
|
Adalimumab
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0348
|
Cladribine
|
C10H12ClN5O3
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0614
|
Etanercept
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0736
|
Golimumab
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0802
|
Infliximab
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0146
|
Baricitinib
|
C16H17N7O2S
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0304
|
Certolizumab pegol
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0661
|
Fingolimod
|
C19H33NO2
|
|
Major | Inter Info |
[26]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1474
|
Siponimod
|
C29H35F3N2O3
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1556
|
Teriflunomide
|
C12H9F3N2O2
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1600
|
Tofacitinib
|
C16H20N6O
|
|
Major | Inter Info |
[34]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1666
|
Upadacitinib
|
C17H19F3N6O
|
|
Major | Inter Info |
[35]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0900
|
Leflunomide
|
C12H9F3N2O2
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1111
|
Natalizumab
|
NA
|
NA
|
Major | Inter Info |
[36]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0032
|
Alefacept
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0033
|
Alemtuzumab
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0085
|
Anakinra
|
C20H23N5O7S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0136
|
Azathioprine
|
C9H7N7O2S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0246
|
Canakinumab
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0313
|
Chloramphenicol
|
C11H12Cl2N2O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0314
|
Chloramphenicol (ophthalmic)
|
C11H12Cl2N2O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0443
|
Denosumab
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0553
|
Efalizumab
|
NA
|
NA
|
Moderate | Inter Info |
[50]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0499
|
Dimethyl fumarate
|
C6H8O4
|
|
Moderate | Inter Info |
[68]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0507
|
Diroximel fumarate
|
C11H13NO6
|
|
Moderate | Inter Info |
[68]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1085
|
Monomethyl fumarate
|
C5H6O4
|
|
Moderate | Inter Info |
[68]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1140
|
Niraparib
|
C19H20N4O
|
|
Moderate | Inter Info |
[70]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1162
|
Ocrelizumab
|
NA
|
NA
|
Moderate | Inter Info |
[71]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1167
|
Olaparib
|
C24H23FN4O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1375
|
Radium Ra 223 dichloride
|
NA
|
NA
|
Moderate | Inter Info |
[73]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1487
|
Sodium phosphate, monobasic (p32)
|
NA
|
NA
|
Moderate | Inter Info |
[76]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1509
|
Strontium chloride Sr-89
|
Cl2Sr
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1174
|
Omacetaxine mepesuccinate
|
C29H39NO9
|
|
Moderate | Inter Info |
[79]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1855
|
Palifermin
|
NA
|
NA
|
Moderate | Inter Info |
[81]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1403
|
Rilonacept
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1422
|
Roflumilast
|
C17H14Cl2F2N2O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1610
|
Topotecan
|
C23H23N3O5
|
|
Moderate | Inter Info |
[87]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1671
|
Ustekinumab
|
NA
|
NA
|
Moderate | Inter Info |
[88]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1720
|
Zidovudine
|
C10H13N5O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Additive myelosuppressive effects | Drug Num: 4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0433
|
Deferiprone
|
C7H9NO2
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1443
|
Samarium (153Sm) lexidronam
|
Sm
|
|
Major | Inter Info |
[37]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0710
|
Ganciclovir
|
C9H13N5O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1676
|
Valganciclovir
|
C14H22N6O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Increased risk of nephrotoxicity | Drug Num: 30 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0335
|
Cidofovir
|
C8H14N3O6P
|
|
Major | Inter Info |
[5]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0432
|
Deferasirox
|
C21H15N3O4
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0472
|
Diatrizoate
|
C11H9I3N2O4
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0629
|
Everolimus
|
C53H83NO14
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0765
|
Human immunoglobulin G (intravenous)
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0761
|
Human botulinum neurotoxin A/B immune globulin
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0763
|
Human cytomegalovirus immune globulin
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0768
|
Human Rho(D) immune globulin
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0805
|
Inotersen
|
C230H318N69O121P19S19
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0834
|
Iopamidol
|
C17H22I3N3O8
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0836
|
Iopromide
|
C18H24I3N3O8
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0837
|
Iothalamic acid
|
C11H9I3N2O4
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0838
|
Ioversol
|
C18H24I3N3O9
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0839
|
Ioxilan
|
C18H24I3N3O8
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0831
|
Iodipamide
|
C20H14I6N2O6
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0832
|
Iodixanol
|
C35H44I6N6O15
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0833
|
Iohexol
|
C19H26I3N3O9
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1476
|
Sirolimus
|
C51H79NO13
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1526
|
Tacrolimus
|
C44H69NO12
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1549
|
Temsirolimus
|
C56H87NO16
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0145
|
Balsalazide
|
C17H15N3O6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0357
|
Clofarabine
|
C10H11ClFN5O3
|
|
Moderate | Inter Info |
[48]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0421
|
Daptomycin
|
C72H101N17O26
|
|
Moderate | Inter Info |
[6]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0783
|
Ibandronate
|
C9H23NO7P2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1009
|
Mesalazine
|
C7H7NO3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1030
|
Methoxyflurane
|
C3H4Cl2F2O
|
|
Moderate | Inter Info |
[6]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1173
|
Olsalazine
|
C14H10N2O6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1217
|
Pamidronic acid
|
C3H11NO7P2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1517
|
Sulfasalazine
|
C18H14N4O5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1727
|
Zoledronic acid
|
C5H10N2O7P2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Increased risk of other adverse reactions (Unspecific) | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0789
|
Idelalisib
|
C22H18FN7O
|
|
Moderate | Inter Info |
[69]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pharmacodynamic antagonistic effects | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antagonize the effect of vaccine/toxoid | Drug Num: 23 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1744
|
Bacillus calmette-guerin substrain tice live antigen
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1839
|
Measles virus vaccine live attenuated
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1849
|
Mumps virus strain B level jeryl lynn live antigen
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1881
|
Rotavirus vaccine
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1882
|
Rubella virus vaccine
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1890
|
Smallpox (Vaccinia) Vaccine, Live
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1897
|
Talimogene laherparepvec
|
NA
|
NA
|
Major | Inter Info |
[31]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1914
|
Typhoid vaccine (live)
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1920
|
Varicella Zoster Vaccine (Recombinant)
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1922
|
Yellow Fever Vaccine
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0089
|
Anthrax vaccine
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0366
|
Clostridium tetani toxoid antigen (formaldehyde inactivated)
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0755
|
Hepatitis A Vaccine
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0756
|
Hepatitis B Vaccine (Recombinant)
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0767
|
Human papillomavirus type 11 L1 capsid protein antigen
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0803
|
Influenza A virus A/California/7/2009 (H1N1)-like antigen (propiolactone inactivated)
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0804
|
Influenza A virus A/Vietnam/1194/2004 (H5N1) antigen (formaldehyde inactivated)
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0999
|
Meningococcal polysaccharide vaccine group C
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1259
|
Pfizer-BioNTech Covid-19 Vaccine
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1307
|
Poliovirus type 1 antigen (formaldehyde inactivated)
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1662
|
Typhoid vaccine (inactivated)
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1692
|
Vibrio cholerae CVD 103-HgR strain live antigen
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1838
|
Lyme disease vaccine (recombinant OspA)
|
NA
|
NA
|
Moderate | Inter Info |
[78]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antagonize the effect of probiotics | Drug Num: 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1749
|
Bifidobacterium longum infantis
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1827
|
Lactobacillus acidophilus
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diminish the response to diagnostic skin test antigens | Drug Num: 4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1755
|
Candida albicans
|
C29H27N2O12P
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1765
|
Coccidioides immitis spherule
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1912
|
Trichophyton mentagrophytes
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1913
|
Tuberculin purified protein derivative
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Attenuated pharmacological effects (Unspecific) | Drug Num: 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0659
|
Filgrastim
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1235
|
Pegfilgrastim
|
C27H46N4O19
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1446
|
Sargramostim
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||