Details of Drug-Drug Interaction
| Drug General Information (ID: DDIY6PX9BO) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Toremifene | Drug Info | Polyethylene glycol (3350) | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Laxatives | |||||||
| Structure | |||||||||
| Mechanism of Toremifene-Polyethylene glycol (3350) Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
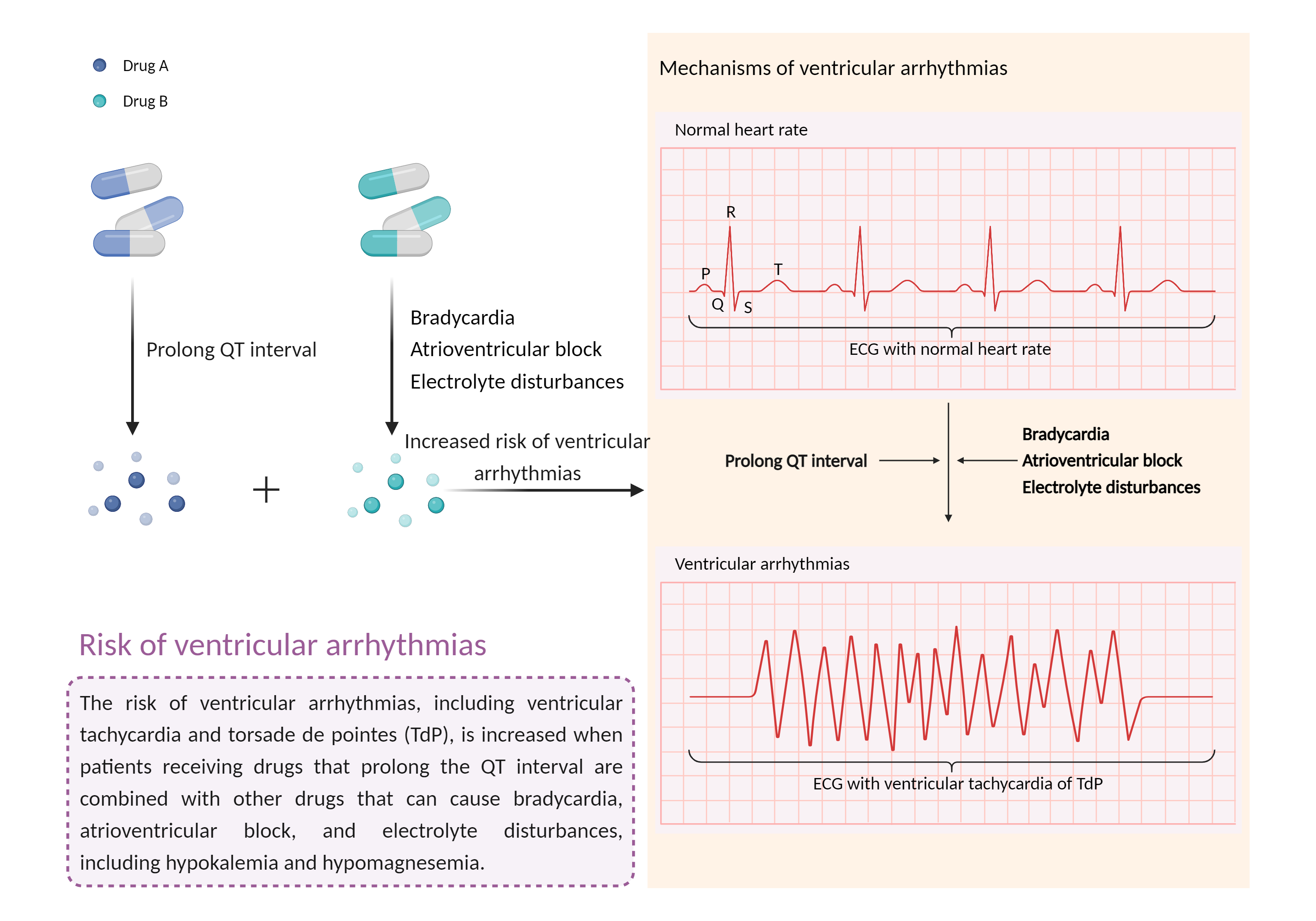
| Increased risk of ventricular arrhythmias Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Toremifene | Polyethylene glycol (3350) | |||||||
| Mechanism | Prolong QT interval | Electrolyte disturbances | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Ventricular arrhythmias | ||||||||
| Factor Description | Ventricular arrhythmias are abnormal heart rhythms that cause your heart's lower chambers to pump blood instead of pumping it. This can limit or stop your heart from supplying blood to your body. While some of these arrhythmias are harmless and do not cause symptoms, others can have serious, even fatal, effects on your body. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Patients treated with drugs that prolong the QT interval should exercise caution when self-medicating with laxatives. The recommended dosage and duration of use should not be exceeded. Patients should be advised to seek prompt medical attention if they experience symptoms that could indicate the occurrence of torsade de pointes such as dizziness, lightheadedness, fainting, palpitation, irregular heart rhythm, shortness of breath, or syncope. | ||||||||
