Details of Drug-Drug Interaction
| Drug General Information (ID: DDIY42DRT6) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Chlorpromazine | Drug Info | Sodium sulfate | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antipsychotic Agents | Laxatives | |||||||
| Structure | |||||||||
| Mechanism of Chlorpromazine-Sodium sulfate Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
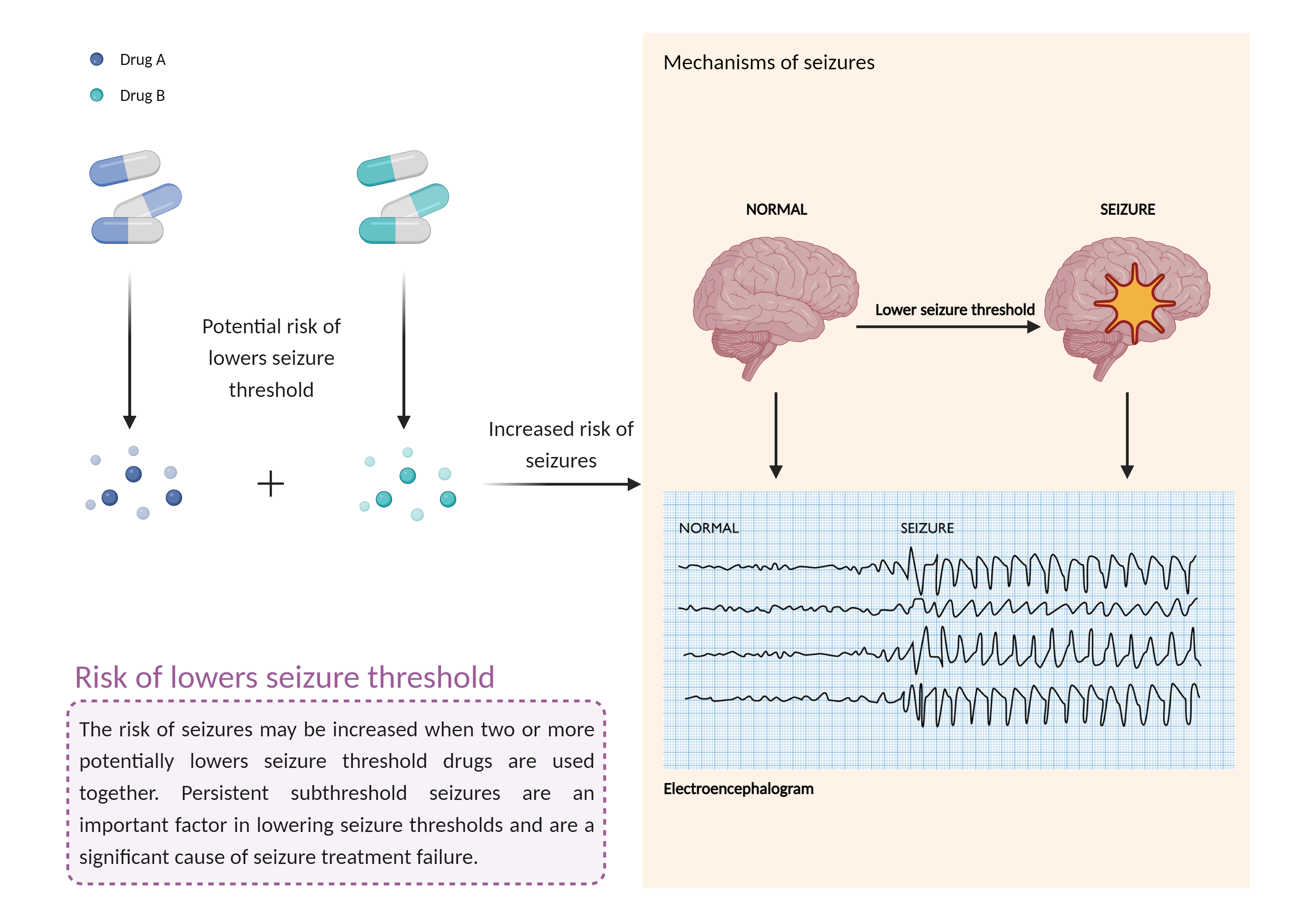
| Increased risk of lowers seizure threshold Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Chlorpromazine | Sodium sulfate | |||||||
| Mechanism 1 | Lower seizure threshold | Lower seizure threshold | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Lowers seizure threshold | ||||||||
| Factor Description | The combination of medications that lower the seizure threshold is a factor that makes people with epilepsy more likely to have seizures. A seizure is a sudden, uncontrolled electrical disturbance in the brain that can cause changes in your behavior, movements or sensations, and level of consciousness. | ||||||||
| Mechanism Description |
|
||||||||
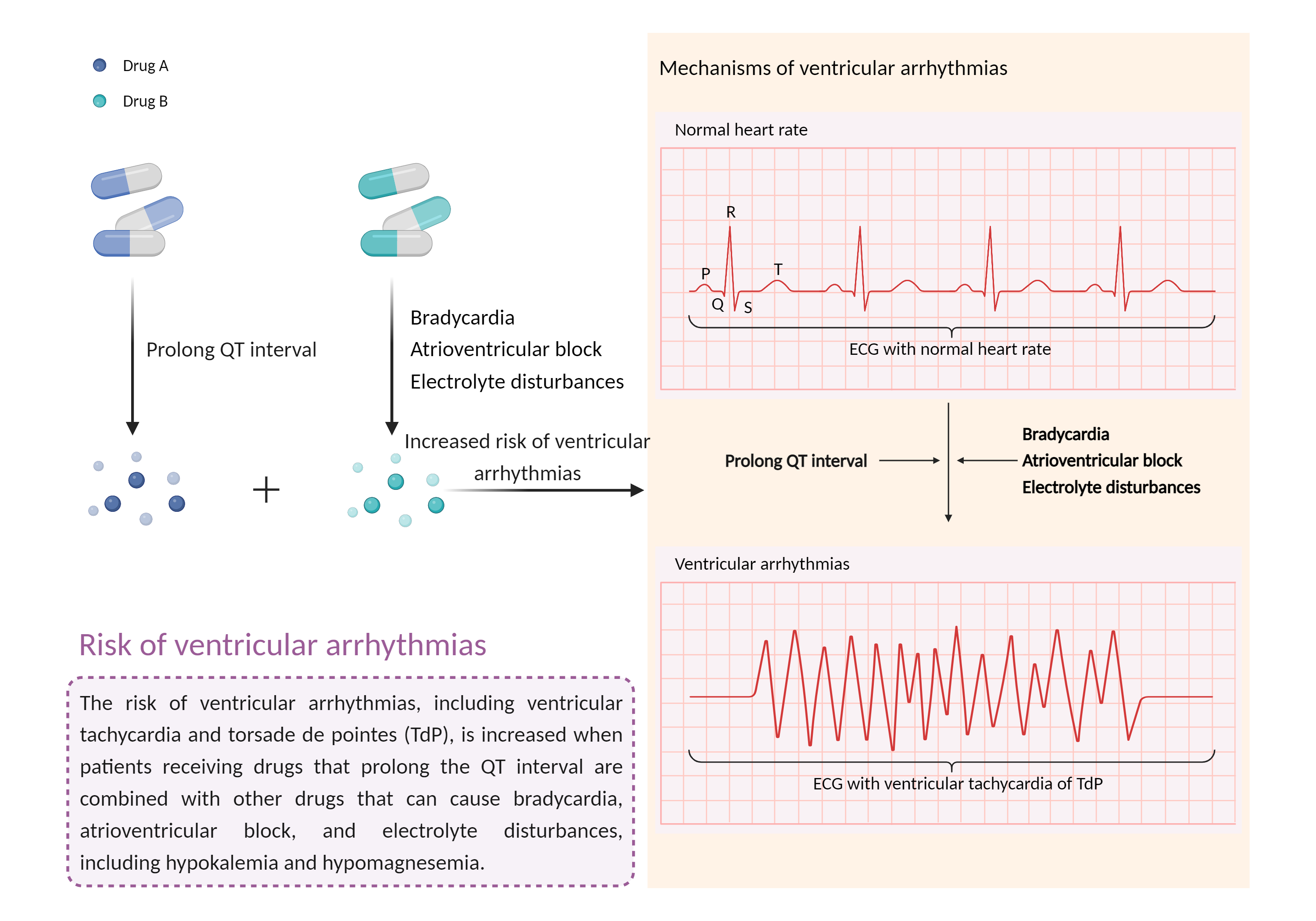
| Increased risk of ventricular arrhythmias Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Chlorpromazine | Sodium sulfate | |||||||
| Mechanism 2 | Prolong QT interval | Electrolyte disturbances | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Ventricular arrhythmias | ||||||||
| Factor Description | Ventricular arrhythmias are abnormal heart rhythms that cause your heart's lower chambers to pump blood instead of pumping it. This can limit or stop your heart from supplying blood to your body. While some of these arrhythmias are harmless and do not cause symptoms, others can have serious, even fatal, effects on your body. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | INTERVAL: Administration of chlorpromazine with Suprep Bowel Prep (magnesium sulfate, potassium sulfate, sodium sulfates) may decrease chlorpromazine serum concentrations. The proposed mechanism is chelation of chlorpromazine by magnesium, forming an insoluble complex that is poorly absorbed from the gastrointestinal tract. If coadministration of chlorpromazine is necessary, administer at least 2 hours before and not less than 6 hours after Suprep Bowel Prep. Caution is advised when bowel cleansing preparations are prescribed in patients treated with agents that can lower the seizure threshold and/or prolong the QT interval. Bowel cleansing preparations should not be used if these patients have impaired renal function or perfusion, dehydration, or uncorrected electrolyte abnormalities. Baseline and postprocedure labs including serum electrolytes, phosphate, BUN, and creatinine should be considered, particularly in the elderly. Patients should be advised not to exceed the recommended dosage of their bowel cleansing preparation and to drink sufficient quantities of clear fluids before, during, and after the bowel preparation process. Administration of an electrolyte rehydration solution may help attenuate the electrolyte abnormalities and hypovolemia. | ||||||||
