Details of Drug-Drug Interaction
| Drug General Information (ID: DDIY0BRFJE) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Deferasirox | Drug Info | Inotersen | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Chelating Agents | Metabolic Agents | |||||||
| Structure | |||||||||
| Mechanism of Deferasirox-Inotersen Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
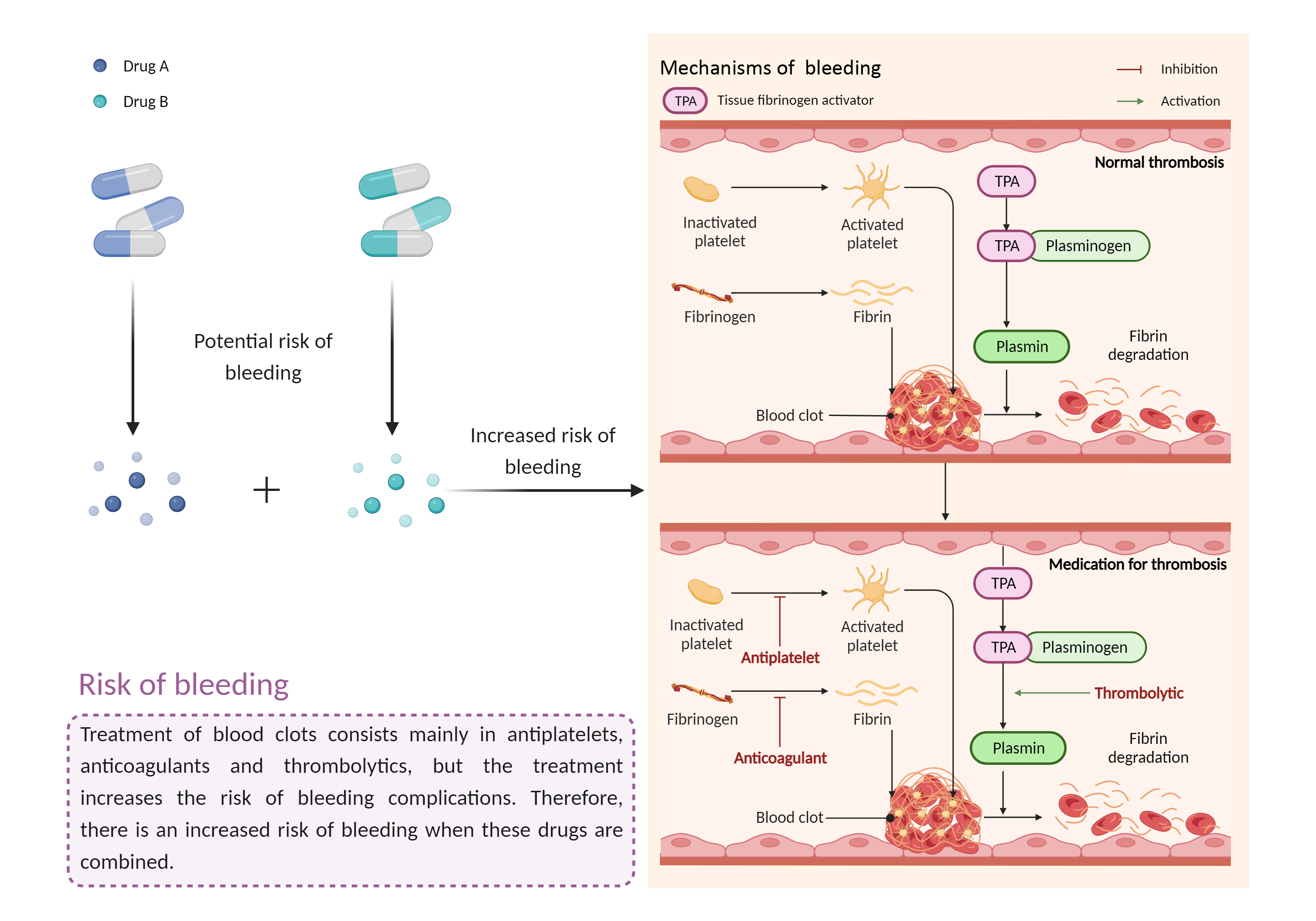
| Increased risk of bleeding Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Deferasirox | Inotersen | |||||||
| Mechanism 1 |
Risk of bleeding Antiplatelet effects |
Risk of bleeding Antiplatelet effects |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Bleeding | ||||||||
| Factor Description | Patients may be at risk for bleeding when the outcome of a disease or disease treatment confounds the standard mechanisms for maintaining hemostasis. Signs or symptoms of abnormal bleeding include: bleeding that takes a long time to stop (including nosebleeds, bleeding gums, bleeding from cuts and abrasions, and menstrual bleeding); severe unexplained bruising, or bruising that becomes larger; blood in the urine or stool, etc. | ||||||||
| Mechanism Description |
|
||||||||
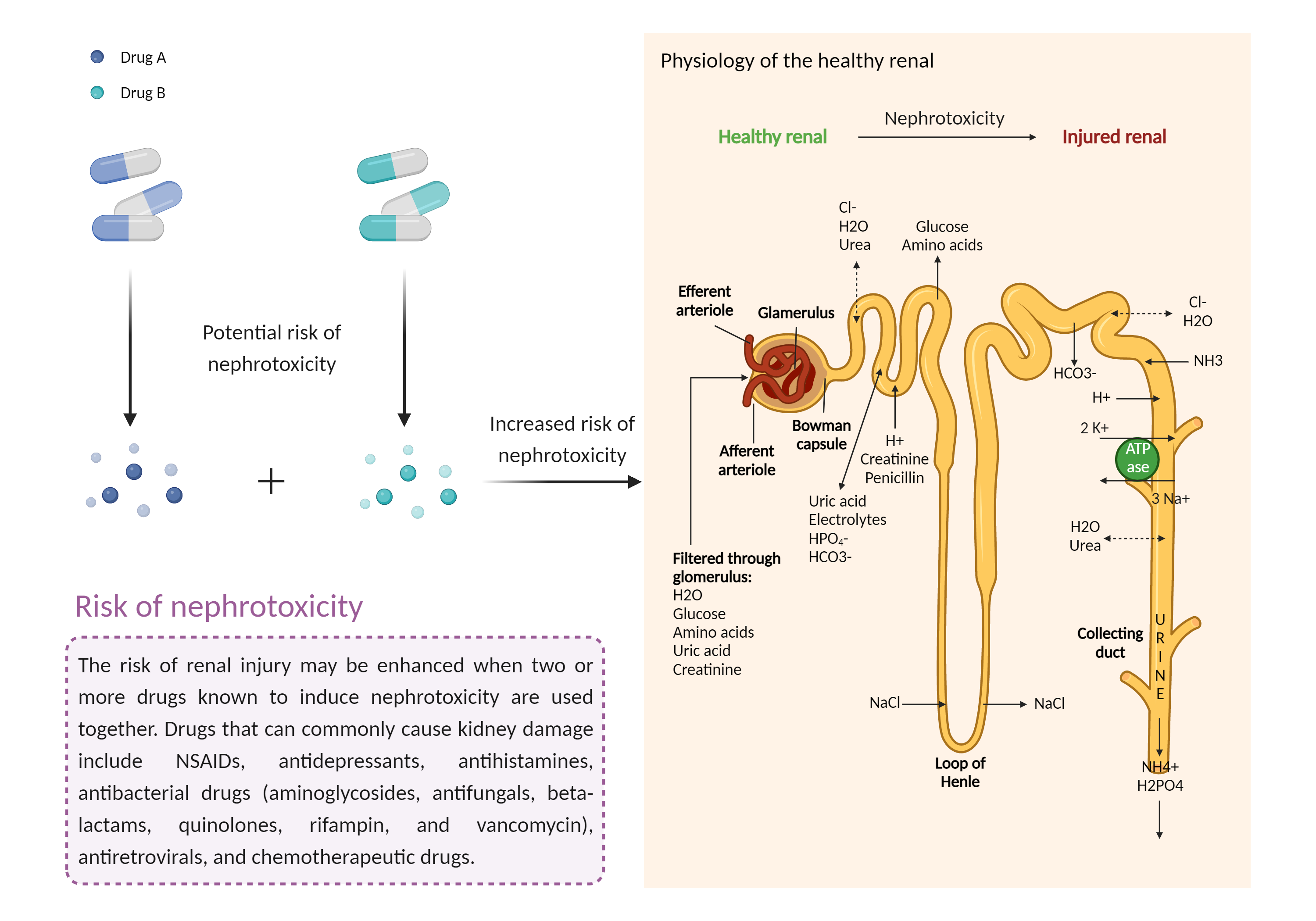
| Increased risk of nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Deferasirox | Inotersen | |||||||
| Mechanism 2 | Nephrotoxicity | Nephrotoxicity | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised when inotersen is prescribed with deferasirox. A platelet count should be obtained prior to initiation of inotersen and regularly during and for at least 8 weeks after treatment in accordance with the product labeling. Inotersen should not be administered in patients with a platelet count below 100 x 10^9/L or in patients who are unable to adhere to the recommended laboratory monitoring and management guidelines. Caution is advised when inotersen is prescribed with other potentially nephrotoxic agents such as deferasirox. Serum creatinine, estimated glomerular filtration rate (eGFR), urine protein to creatinine ratio (UPCR), and a urinalysis should be obtained prior to initiation of inotersen and regularly during and for at least 8 weeks after treatment in accordance with the product labeling. Inotersen should generally not be initiated in patients with a UPCR of 1000 mg/g or higher, or in patients who are unable to adhere to the recommended laboratory monitoring and management guidelines. Patients or their caregivers should be apprised of the signs and symptoms of glomerulonephritis and to seek medical attention if they occur, including edema, shortness of breath, coughing,hematuria, and decreased urination. In patients with UPCR of 2000 mg/g or higher, perform further evaluation for acute glomerulonephritis as clinically indicated. If acute glomerulonephritis is confirmed, inotersen should be permanently discontinued. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
| 2 | Product Information. Tegsedi (inotersen). Akcea Therapeutics, Cambridge, MA. | ||||||||||||||||||
