Details of Drug-Drug Interaction
| Drug General Information (ID: DDIX9RK58A) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Ribavirin | Drug Info | Azathioprine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiviral Agents | Immunosuppressive Agents | |||||||
| Structure | |||||||||
| Mechanism of Ribavirin-Azathioprine Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Non-CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
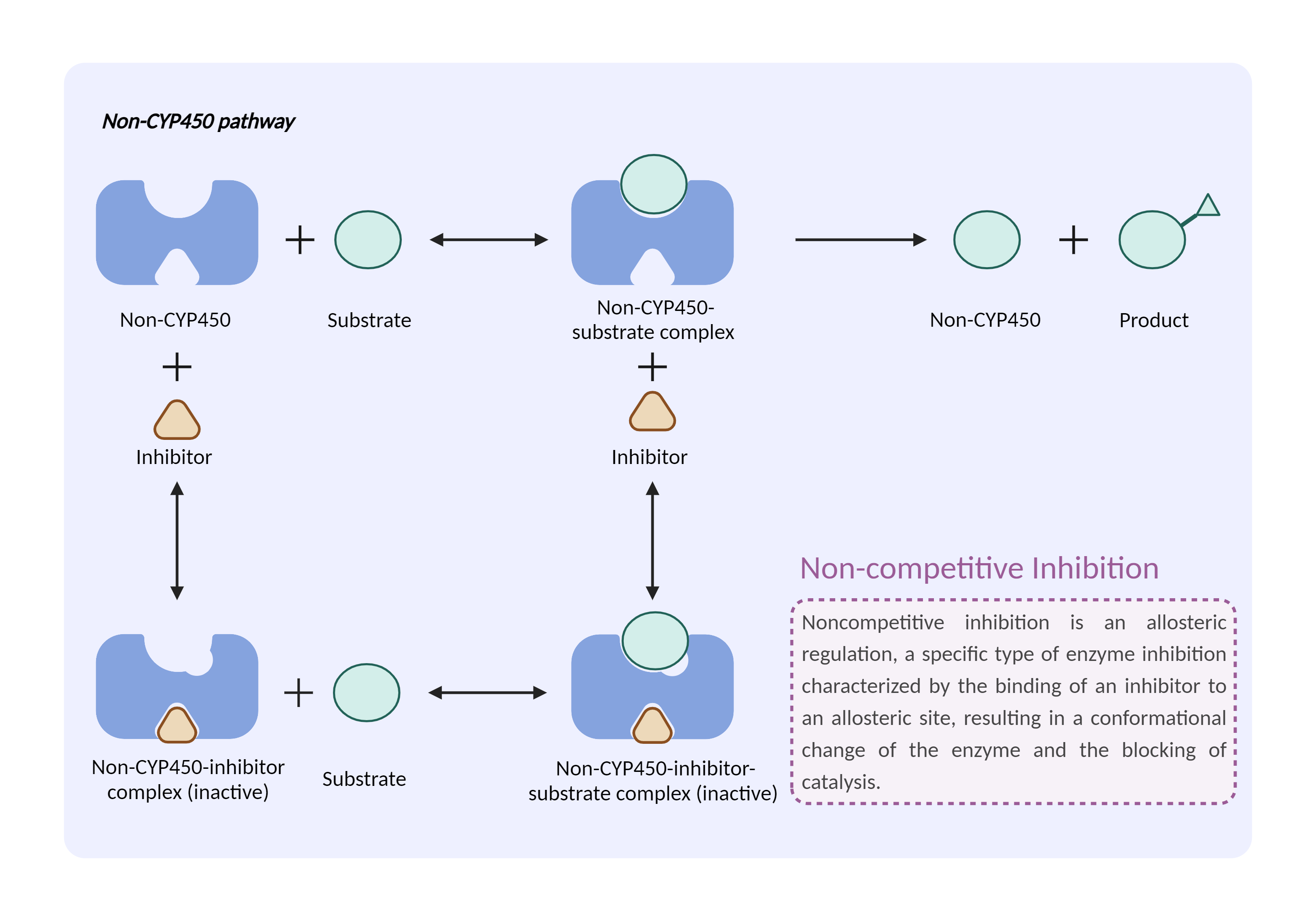 |
|||||||||
| Drug Name | Ribavirin | Azathioprine | |||||||
| Mechanism | Inosine monophosphate dehydrogenase inhibitor | Inosine monophosphate dehydrogenase substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Inosine-5'-monophosphate dehydrogenase 1 |
×
Structure
Sequence
MADYLISGGTGYVPEDGLTAQQLFASADGLTYNDFLILPGFIDFIADEVDLTSALTRKITLKTPLISSPMDTVTEADMAIAMALMGGIGFIHHNCTPEFQANEVRKVKKFEQGFITDPVVLSPSHTVGDVLEAKMRHGFSGIPITETGTMGSKLVGIVTSRDIDFLAEKDHTTLLSEVMTPRIELVVAPAGVTLKEANEILQRSKKGKLPIVNDCDELVAIIARTDLKKNRDYPLASKDSQKQLLCGAAVGTREDDKYRLDLLTQAGVDVIVLDSSQGNSVYQIAMVHYIKQKYPHLQVIGGNVVTAAQAKNLIDAGVDGLRVGMGCGSICITQEVMACGRPQGTAVYKVAEYARRFGVPIIADGGIQTVGHVVKALALGASTVMMGSLLAATTEAPGEYFFSDGVRLKKYRGMGSLDAMEKSSSSQKRYFSEGDKVKIAQGVSGSIQDKGSIQKFVPYLIAGIQHGCQDIGARSLSVLRSMMYSGELKFEKRTMSAQIEGGVHGLHSYEKRLY
|
|||||||
| Gene Name | IMPDH1 | ||||||||
| Uniprot ID | IMDH1_HUMAN | ||||||||
| KEGG Pathway | hsa:3614 | ||||||||
| Protein Family | IMPDH/GMPR family | ||||||||
| Protein Function |
Catalyzes the conversion of inosine 5'-phosphate (IMP) to xanthosine 5'-phosphate (XMP), the first committed and rate-limiting step in the de novo synthesis of guanine nucleotides, and therefore plays an important role in the regulation of cell growth. Could also have a single-stranded nucleic acid-binding activity and could play a role in RNA and/or DNA metabolism. It may also have a role in the development of malignancy and the growth progression of some tumors.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The use of ribavirin in combination with 6-mercaptopurine or azathioprine should generally be avoided if possible. Patients receiving the combination should have complete blood counts, including platelet counts, monitored weekly for the first month, twice monthly for the second and third months, then monthly or more frequently if dosage or other therapy changes are necessary. Treatment with these medications should be discontinued promptly if pancytopenia develops, and the combination should not be reintroduced following recovery. | ||||||||
