Details of Drug-Drug Interaction
| Drug General Information (ID: DDIX4FS6ZO) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Sotalol | Drug Info | Gadobutrol | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiarrhythmic Agents | Diagnostic Agents | |||||||
| Structure | |||||||||
| Mechanism of Sotalol-Gadobutrol Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
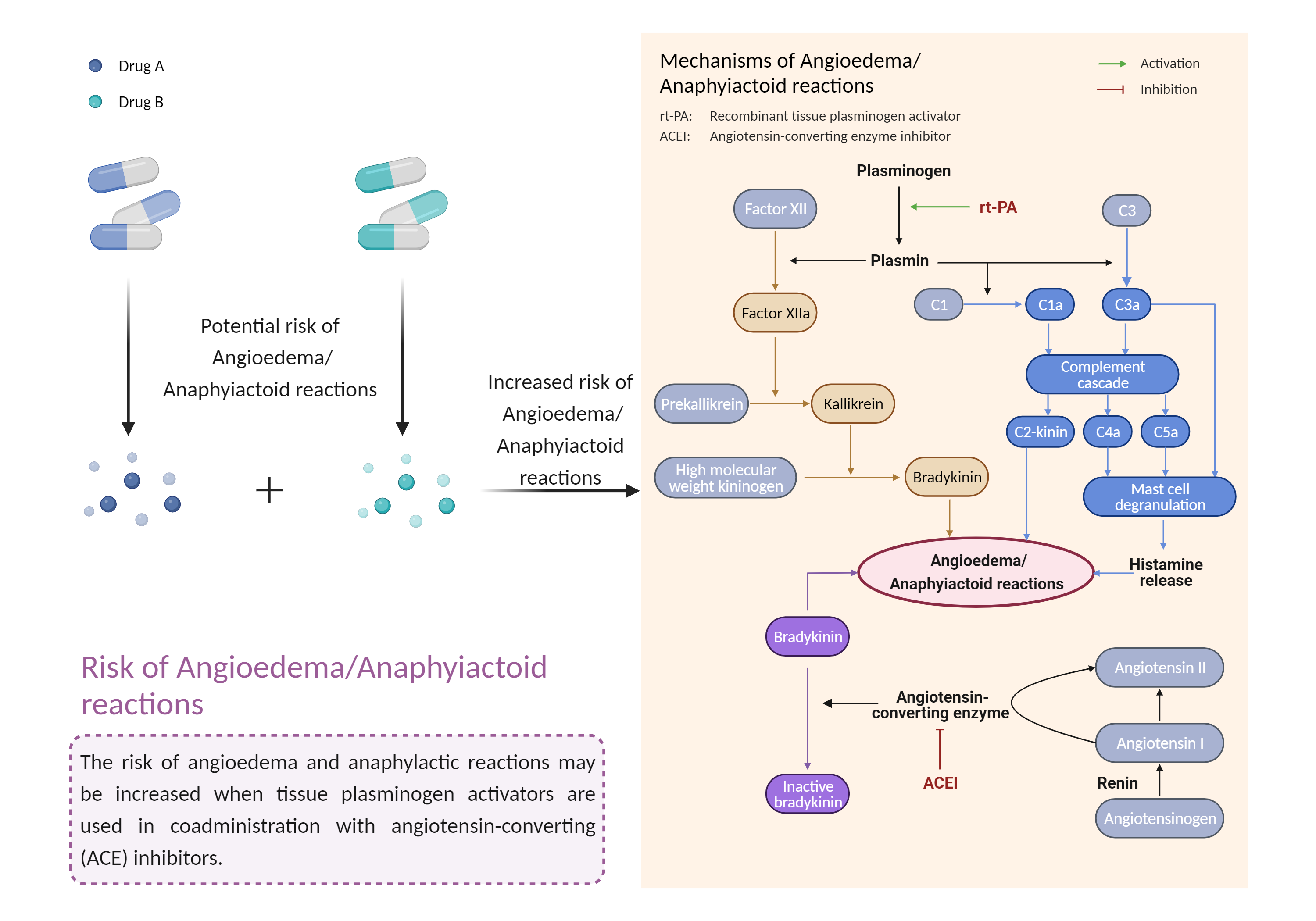
| Increased risk of angioedema/anaphylactoid reactions Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Sotalol | Gadobutrol | |||||||
| Mechanism | Anaphylactoid reactions | Anaphylactoid reactions | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Angioedema/anaphylactoid reactions | ||||||||
| Factor Description | Anaphylactoid reaction is a condition in which symptoms similar to an allergic reaction occur, but no immune sensitization is detected. Most commonly, the syndrome begins with skin involvement, usually pruritus, flushing, urticaria, or angioedema. Angioedema is a reaction to a trigger that causes swelling of the tissue below the inner layer of the skin, called the dermis or submucosa, and the swelling can occur on the face, throat, or around the genitals. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Patients who have received beta-blockers should be closely monitored for adverse reactions to contrast media. If anaphylaxis occurs, clinicians should be aware that beta-blockers may attenuate the response to epinephrine. Thus, larger doses of epinephrine may be necessary to overcome the bronchospasm, although such large doses can also cause excessive alpha-adrenergic stimulation resulting in hypertension, reflex bradycardia, heart block, and possible potentiation of bronchospasm. alternative treatments recommended include vigorous supportive care (e.g., fluids) and the use of parenteral beta-agonists for bronchospasm and norepinephrine for hypotension. | ||||||||
