Details of Drug-Drug Interaction
| Drug General Information (ID: DDIWVCSPOD) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Drotrecogin alfa | Drug Info | Temsirolimus | Drug Info | |||||
| Drug Type | Protein/peptide | Small molecule | |||||||
| Therapeutic Class | Coagulation Modifiers | Antineoplastics/Mtor Inhibitors | |||||||
| Mechanism of Drotrecogin alfa-Temsirolimus Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Increased risk of bleeding Click to Show/Hide Mechanism Graph | |||||||||
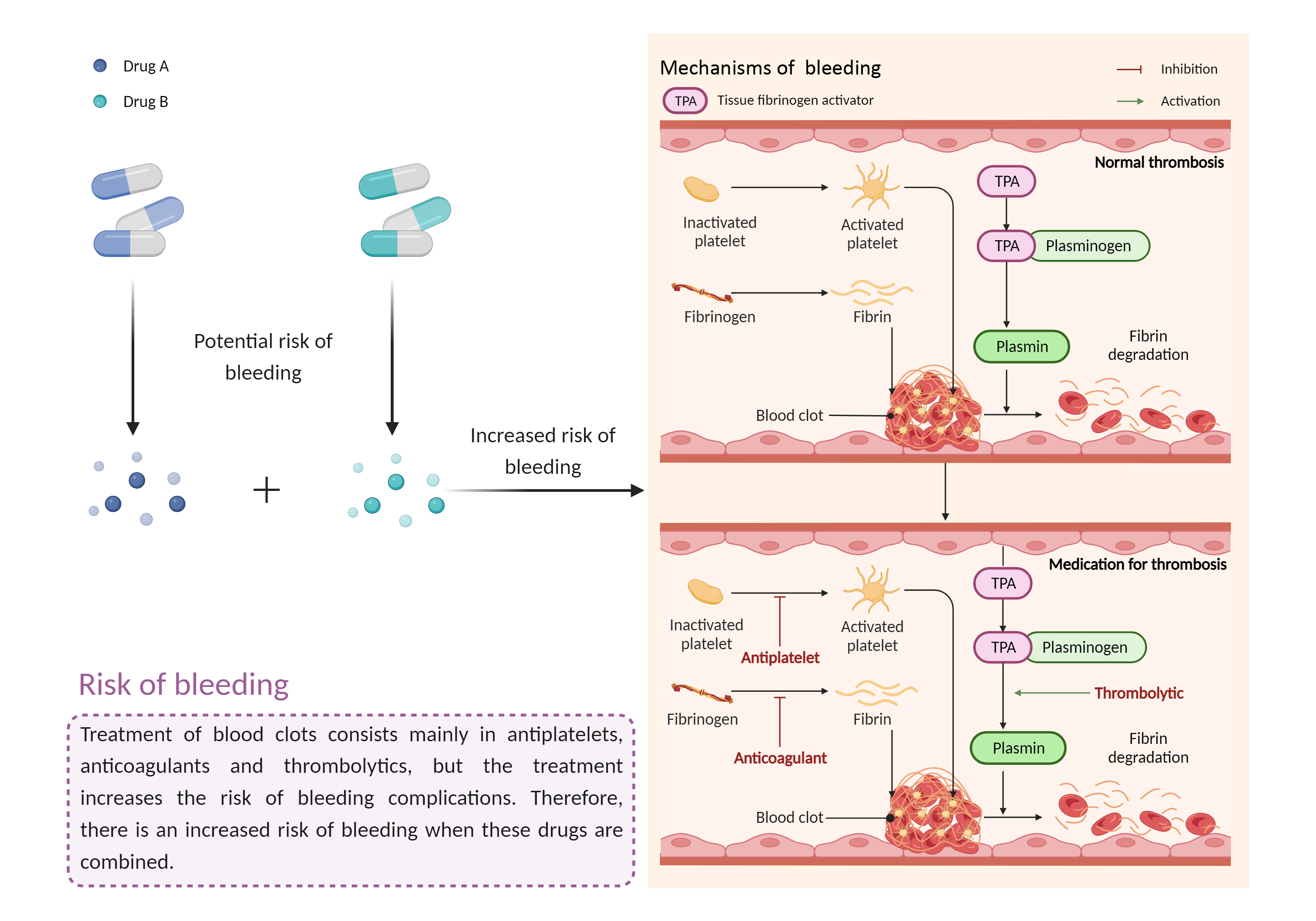 |
|||||||||
| Drug Name | Drotrecogin alfa | Temsirolimus | |||||||
| Mechanism |
Risk of bleeding Anticoagulant effects |
Risk of bleeding Antiplatelet/anticoagulant effects |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Bleeding | ||||||||
| Factor Description | Patients may be at risk for bleeding when the outcome of a disease or disease treatment confounds the standard mechanisms for maintaining hemostasis. Signs or symptoms of abnormal bleeding include: bleeding that takes a long time to stop (including nosebleeds, bleeding gums, bleeding from cuts and abrasions, and menstrual bleeding); severe unexplained bruising, or bruising that becomes larger; blood in the urine or stool, etc. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Patients receiving anticoagulants should be informed of the increased risk of developing intracerebral bleeding while on temsirolimus. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Torisel (temsirolimus). Wyeth-Ayerst Laboratories, Philadelphia, PA. | ||||||||||||||||||
