Details of Drug-Drug Interaction
| Drug General Information (ID: DDIVOJFWD2) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Halothane | Drug Info | Prasugrel | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anesthetics | Antiplatelet Agents | |||||||
| Structure | |||||||||
| Mechanism of Halothane-Prasugrel Interaction (Severity Level: Minor) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
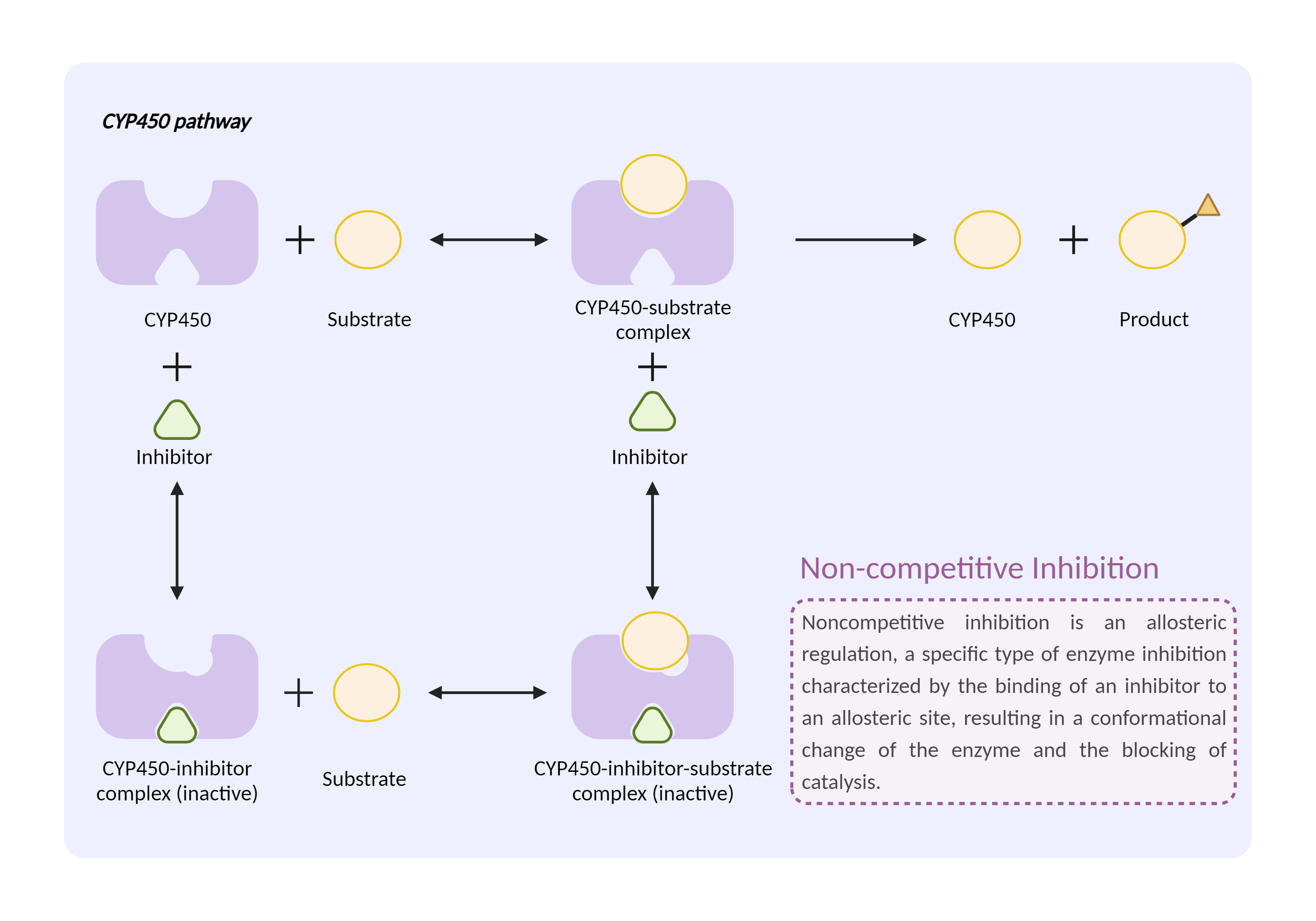 |
|||||||||
| Drug Name | Halothane | Prasugrel | |||||||
| Mechanism | CYP450 2B6 substrate | CYP450 2B6 inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2B6 |
×
Structure
Sequence
MELSVLLFLALLTGLLLLLVQRHPNTHDRLPPGPRPLPLLGNLLQMDRRGLLKSFLRFREKYGDVFTVHLGPRPVVMLCGVEAIREALVDKAEAFSGRGKIAMVDPFFRGYGVIFANGNRWKVLRRFSVTTMRDFGMGKRSVEERIQEEAQCLIEELRKSKGALMDPTFLFQSITANIICSIVFGKRFHYQDQEFLKMLNLFYQTFSLISSVFGQLFELFSGFLKYFPGAHRQVYKNLQEINAYIGHSVEKHRETLDPSAPKDLIDTYLLHMEKEKSNAHSEFSHQNLNLNTLSLFFAGTETTSTTLRYGFLLMLKYPHVAERVYREIEQVIGPHRPPELHDRAKMPYTEAVIYEIQRFSDLLPMGVPHIVTQHTSFRGYIIPKDTEVFLILSTALHDPHYFEKPDAFNPDHFLDANGALKKTEAFIPFSLGKRICLGEGIARAELFLFFTTILQNFSMASPVAPEDIDLTPQECGVGKIPPTYQIRFLPR
|
|||||||
| Gene Name | CYP2B6 | ||||||||
| Uniprot ID | CP2B6_HUMAN | ||||||||
| KEGG Pathway | hsa:1555 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of endocannabinoids and steroids (PubMed:21289075, PubMed:12865317). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase). Catalyzes the epoxidation of double bonds of arachidonoylethanolamide (anandamide) to 8,9-, 11,12-, and 14,15-epoxyeicosatrienoic acid ethanolamides (EpETrE-EAs), potentially modulating endocannabinoid system signaling (PubMed:21289075). Hydroxylates steroid hormones, including testosterone at C-16 and estrogens at C-2 (PubMed:21289075, PubMed:12865317). Plays a role in the oxidative metabolism of xenobiotics, including plant lipids and drugs (PubMed:11695850, PubMed:22909231). Acts as a 1,4-cineole 2-exo-monooxygenase (PubMed:11695850).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
| 2 | Product Information. Effient (prasugrel). Lilly, Eli and Company, Indianapolis, IN. | ||||||||||||||||||
