Details of Drug-Drug Interaction
| Drug General Information (ID: DDIVIAZSWC) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Lyme disease vaccine (recombinant OspA) | Drug Info | Lutetium Lu 177 dotatate | Drug Info | |||||
| Drug Type | Vaccine | Small molecule | |||||||
| Therapeutic Class | Vaccine | Therapeutic Radiopharmaceuticals | |||||||
| Mechanism of Lyme disease vaccine (recombinant OspA)-Lutetium Lu 177 dotatate Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
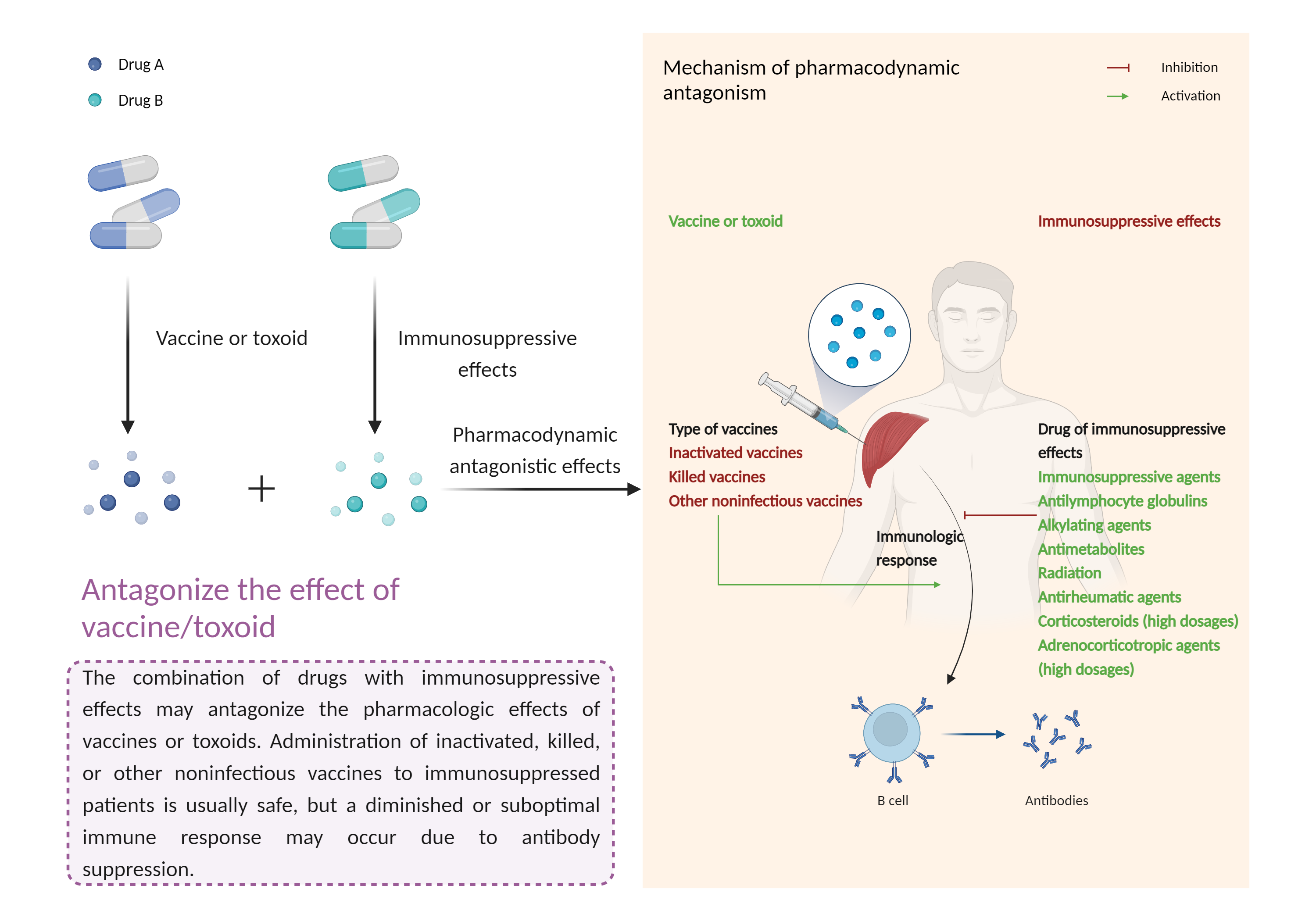
| Antagonize the effect of vaccine/toxoid Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Lyme disease vaccine (recombinant OspA) | Lutetium Lu 177 dotatate | |||||||
| Mechanism | Vaccine or toxoid | Immunosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Vaccine/toxoid | ||||||||
| Factor Description | The beneficial effects of the vaccine/toxinoid may be reduced, leading to a diminished or suboptimal immune response. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The manufacturer suggests considering deferral of vaccination against Lyme disease until 3 months after discontinuation of immunosuppressive therapy. Vaccines may generally be administered to patients receiving corticosteroids as replacement therapy (e.g., for Addison's disease). | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Shen AK, Mead PS, Beard CB. The Lyme disease vaccine--a public health perspective. Clin Infect Dis. 2011;52 Suppl 3:s247-s252. [PMID: 21217171] | ||||||||||||||||||
