| Mechanism of Amisulpride-Polyethylene glycol (3350 with electrolytes) Interaction
(Severity Level: Moderate)
|
|
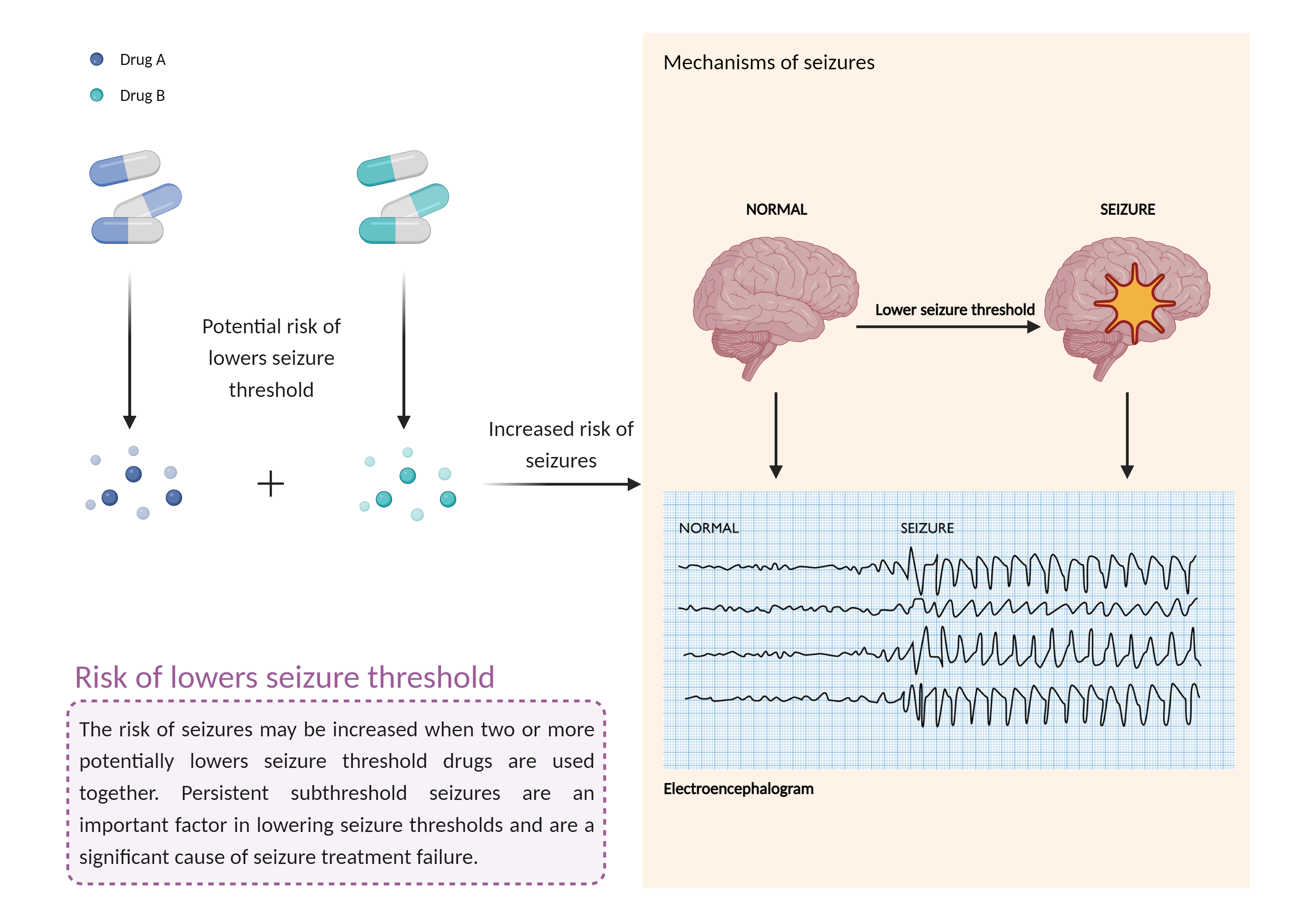
Increased risk of lowers seizure threshold
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Amisulpride |
Polyethylene glycol (3350 with electrolytes) |
|
Mechanism 1
|
Lower seizure threshold |
Lower seizure threshold |
| Key Mechanism Factor 1 |
| Factor Name |
Lowers seizure threshold |
| Factor Description |
The combination of medications that lower the seizure threshold is a factor that makes people with epilepsy more likely to have seizures. A seizure is a sudden, uncontrolled electrical disturbance in the brain that can cause changes in your behavior, movements or sensations, and level of consciousness. |
| Mechanism Description |
- Increased risk of lowers seizure threshold by the combination of Amisulpride and Polyethylene glycol (3350 with electrolytes)
|
|
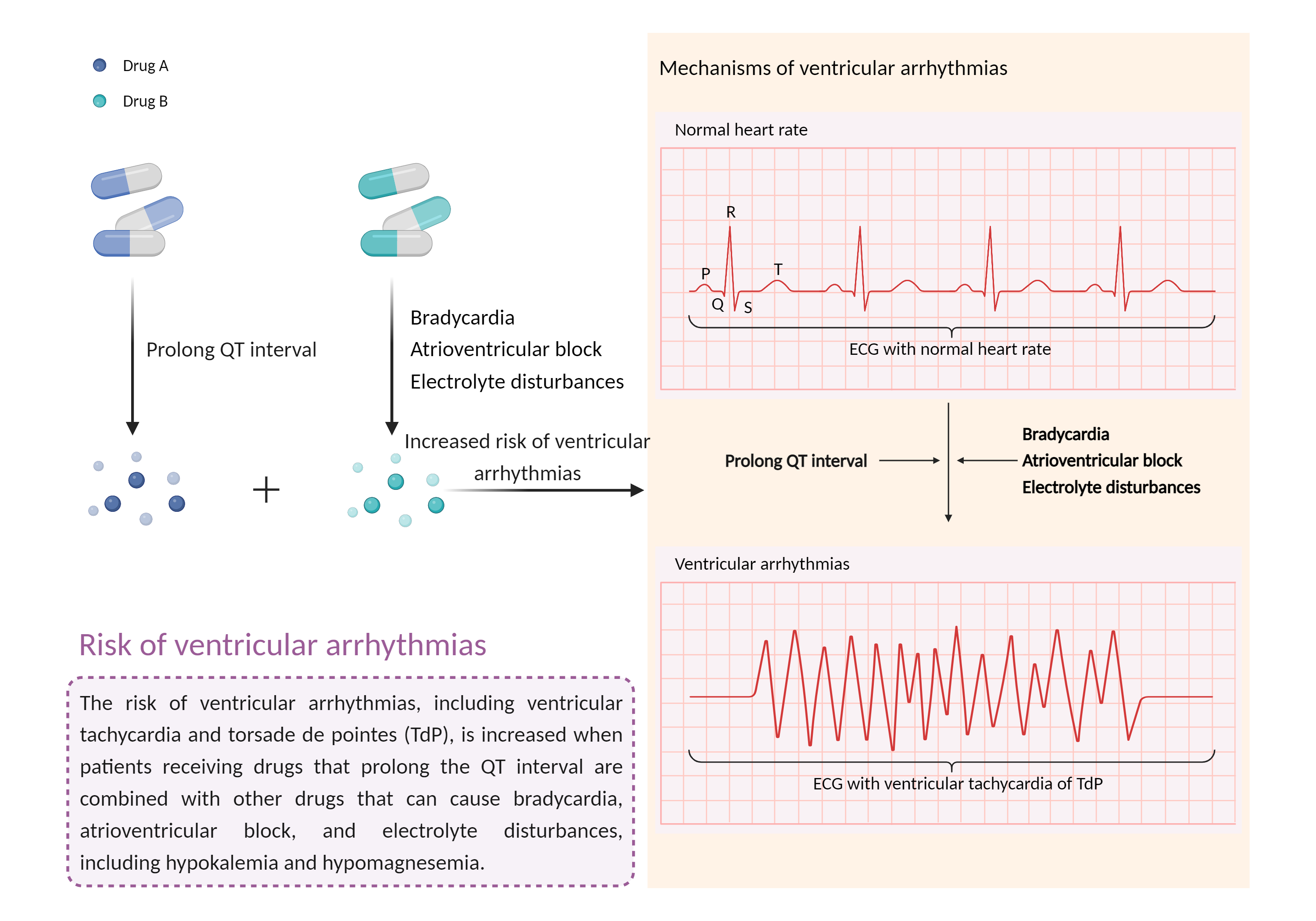
Increased risk of ventricular arrhythmias
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Amisulpride |
Polyethylene glycol (3350 with electrolytes) |
|
Mechanism 2
|
Prolong QT interval |
Electrolyte disturbances |
| Key Mechanism Factor 2 |
| Factor Name |
Ventricular arrhythmias |
| Factor Description |
Ventricular arrhythmias are abnormal heart rhythms that cause your heart's lower chambers to pump blood instead of pumping it. This can limit or stop your heart from supplying blood to your body. While some of these arrhythmias are harmless and do not cause symptoms, others can have serious, even fatal, effects on your body. |
| Mechanism Description |
- Increased risk of ventricular arrhythmias by the combination of Amisulpride and Polyethylene glycol (3350 with electrolytes)
|


