Details of Drug-Drug Interaction
| Drug General Information (ID: DDIVHTC0NG) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Dicoumarol | Drug Info | Ribavirin | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anticoagulants | Antiviral Agents | |||||||
| Structure | |||||||||
| Mechanism of Dicoumarol-Ribavirin Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
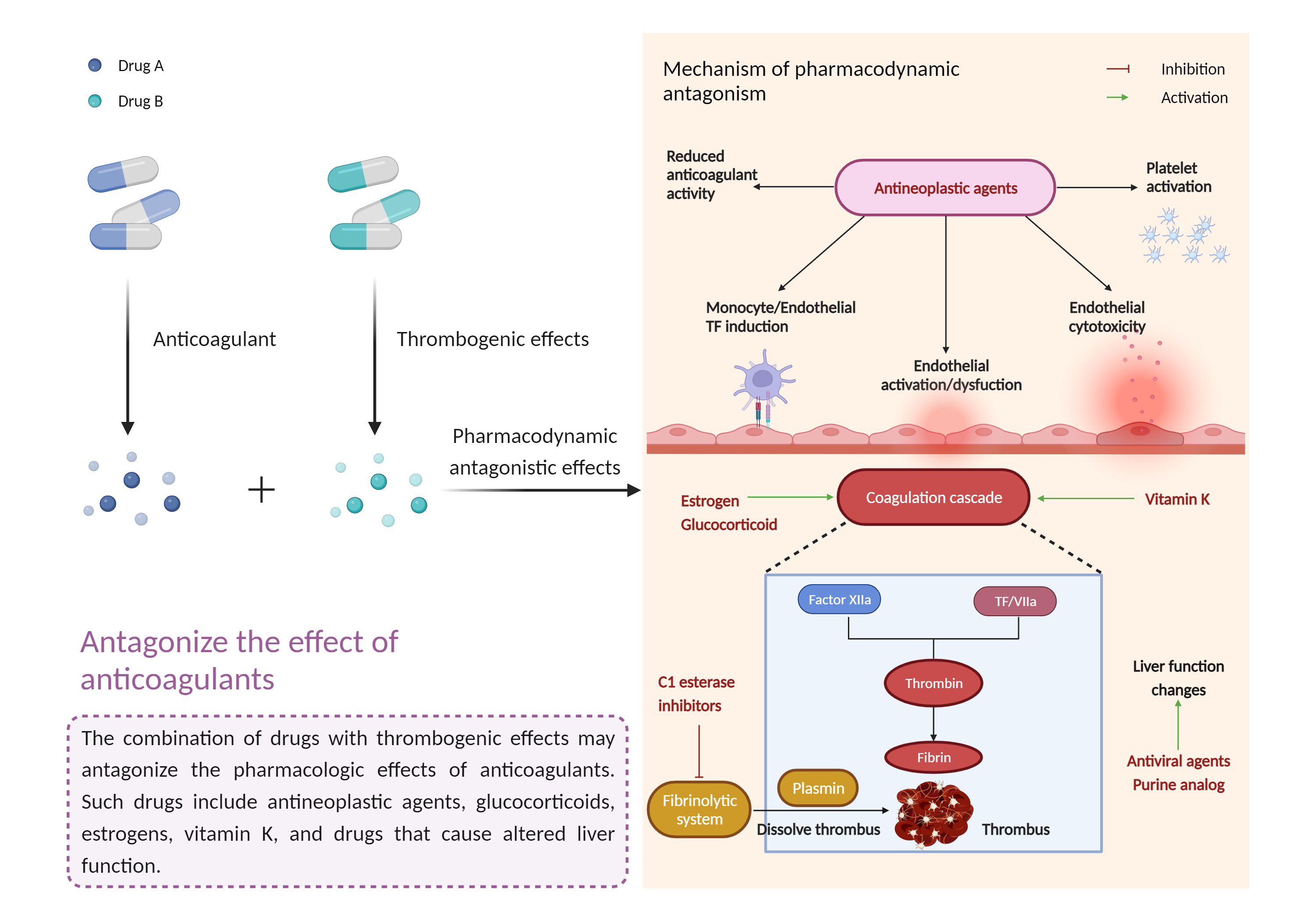
| Antagonize the effect of antithrombotic agents Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Dicoumarol | Ribavirin | |||||||
| Mechanism | Vitamin K antagonist | Decrease the hypoprothrombinemic effect of vitamin K antagonist | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Antithrombotic agents | ||||||||
| Factor Description | The beneficial effects of antithrombotic agents are reduced, leading to an increased risk of thromboembolism, stroke and/or myocardial infarction. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Patients treated with warfarin should be closely monitored during concomitant therapy with ribavirin. The INR should be checked frequently (e.g., weekly) during the 4 weeks following initiation or discontinuation of ribavirin therapy in patients who are stabilized on their warfarin regimen, and the warfarin dosage adjusted accordingly. The same precaution may be applicable during therapy with other oral anticoagulants, although clinical data are lacking. Patients should be advised to promptly report any signs of bleeding to their physician, including pain, swelling, headache, dizziness, weakness, prolonged bleeding from cuts, increased menstrual flow, vaginal bleeding, nosebleeds, bleeding of gums from brushing, unusual bleeding or bruising, red or brown urine, or red or black stools. | ||||||||
