Details of Drug-Drug Interaction
| Drug General Information (ID: DDIVFXC5BH) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Isosorbide mononitrate | Drug Info | Amifostine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Vasodilator Agents | Cytoprotective Agent | |||||||
| Structure | |||||||||
| Mechanism of Isosorbide mononitrate-Amifostine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
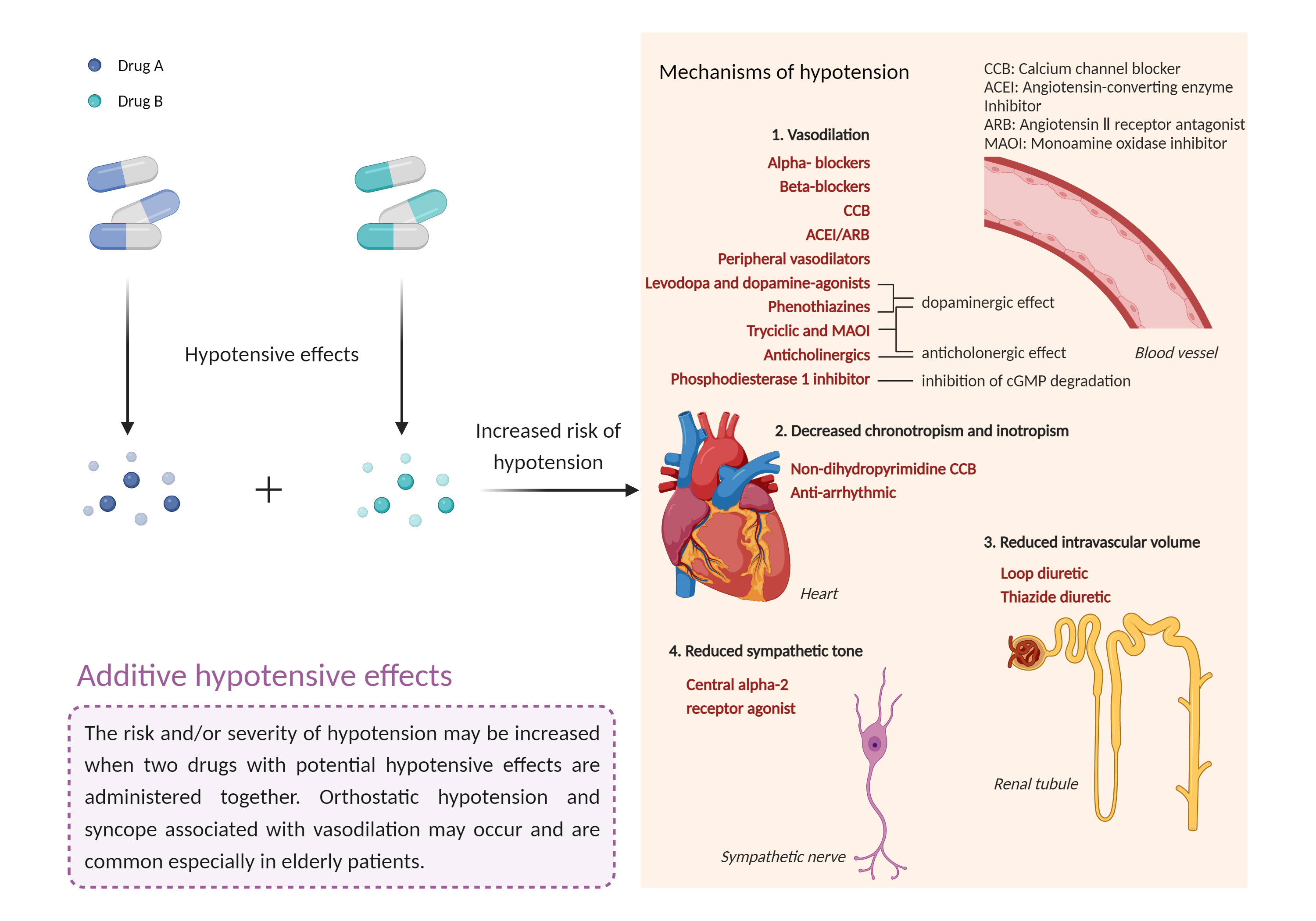 |
|||||||||
| Drug Name | Isosorbide mononitrate | Amifostine | |||||||
| Mechanism 1 |
Hypotensive effects Guanylate cyclase Agonist |
Antihypertensive agent | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Guanylate cyclase soluble | Structure Sequence | |||||||
| Protein Family | Adenylyl cyclase class-4/guanylyl cyclase family | ||||||||
| Protein Function |
There are two types of guanylate cyclases: soluble forms and membrane-associated receptor forms. Activated by nitric oxide in the presence of magnesium or manganese ions.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Mechanism 2 |
Hypotensive effects Guanylate cyclase Agonist |
Hypotensive effects | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Guanylate cyclase soluble | Structure Sequence | |||||||
| Protein Family | Adenylyl cyclase class-4/guanylyl cyclase family | ||||||||
| Protein Function |
There are two types of guanylate cyclases: soluble forms and membrane-associated receptor forms. Activated by nitric oxide in the presence of magnesium or manganese ions.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | If medically feasible, antihypertensive medications should be interrupted 24 hours prior to amifostine administration. Blood pressure should be closely monitored during and after treatment. In addition to hypotension, transient hypertension or exacerbation of preexisting hypertension may occur from intravenous hydration, discontinuation of antihypertensive medications, or other causes. Patients who continue their hypotensive medication(s) during amifostine therapy should be carefully monitored for the development of hypotension. | ||||||||
