| Mechanism of Porfimer sodium-Panitumumab Interaction
(Severity Level: Moderate)
|
|
Increased risk of photosensitivity reactions
Click to Show/Hide Mechanism Graph
|
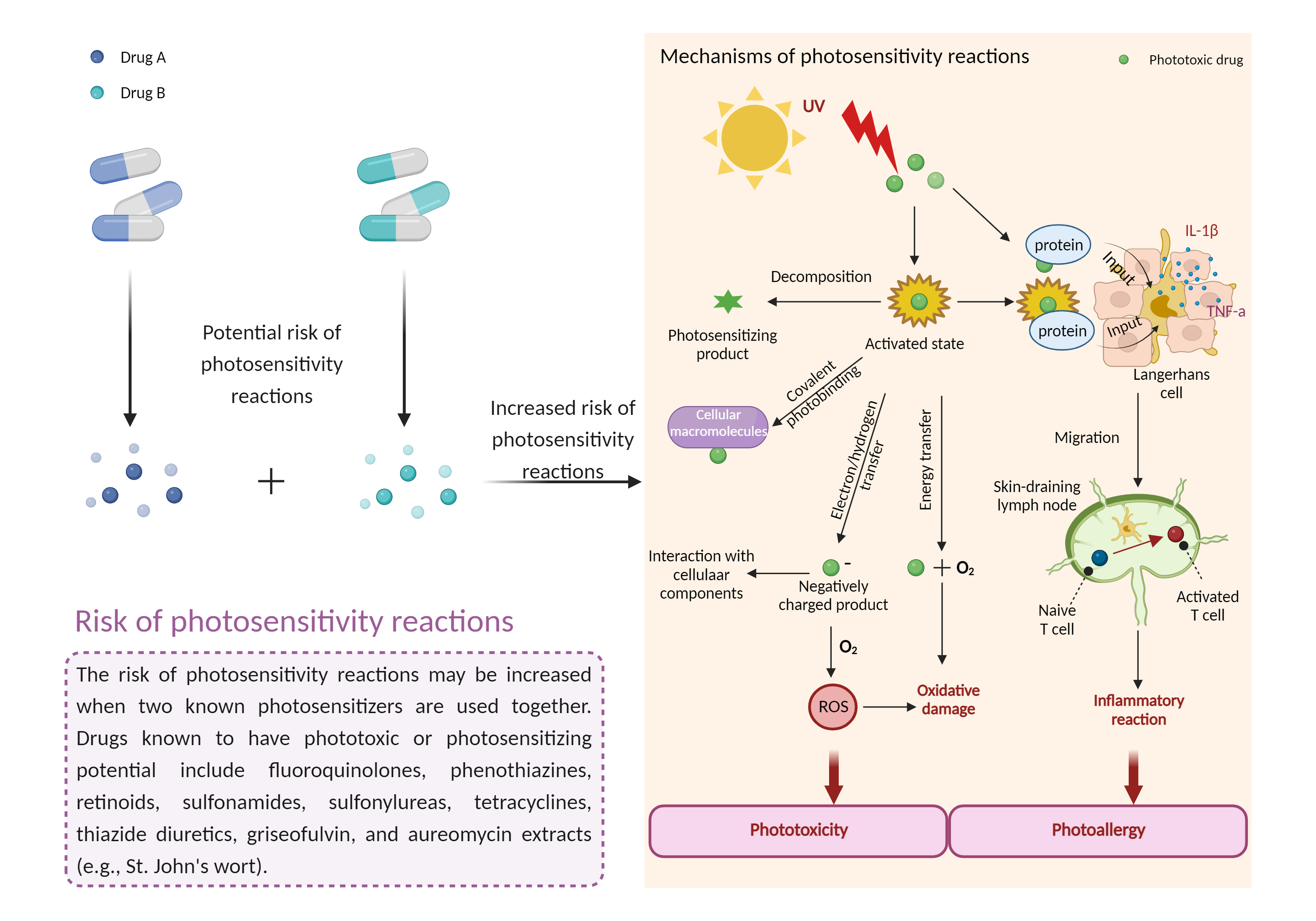 |
| Drug Name |
Porfimer sodium |
Panitumumab |
|
Mechanism
|
Photosensitivity reactions |
Photosensitivity reactions |
| Key Mechanism Factor 1 |
| Factor Name |
Photosensitivity reactions |
| Factor Description |
Drug-induced photosensitivity reactions occur when certain photosensitizing drugs cause unexpected sunburn or dermatitis (dry, bumpy or blistering rash) on sun-exposed skin (face, neck, arms, backs of hands, and often lower legs and feet). The rash may or may not be itchy. |
| Mechanism Description |
- Increased risk of photosensitivity reactions by the combination of Porfimer sodium and Panitumumab
|

