Details of Drug-Drug Interaction
| Drug General Information (ID: DDIV5YNP7S) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Anisindione | Drug Info | Cefepime | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anticoagulants | Antibiotics | |||||||
| Structure | |||||||||
| Mechanism of Anisindione-Cefepime Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Increased risk of bleeding Click to Show/Hide Mechanism Graph | |||||||||
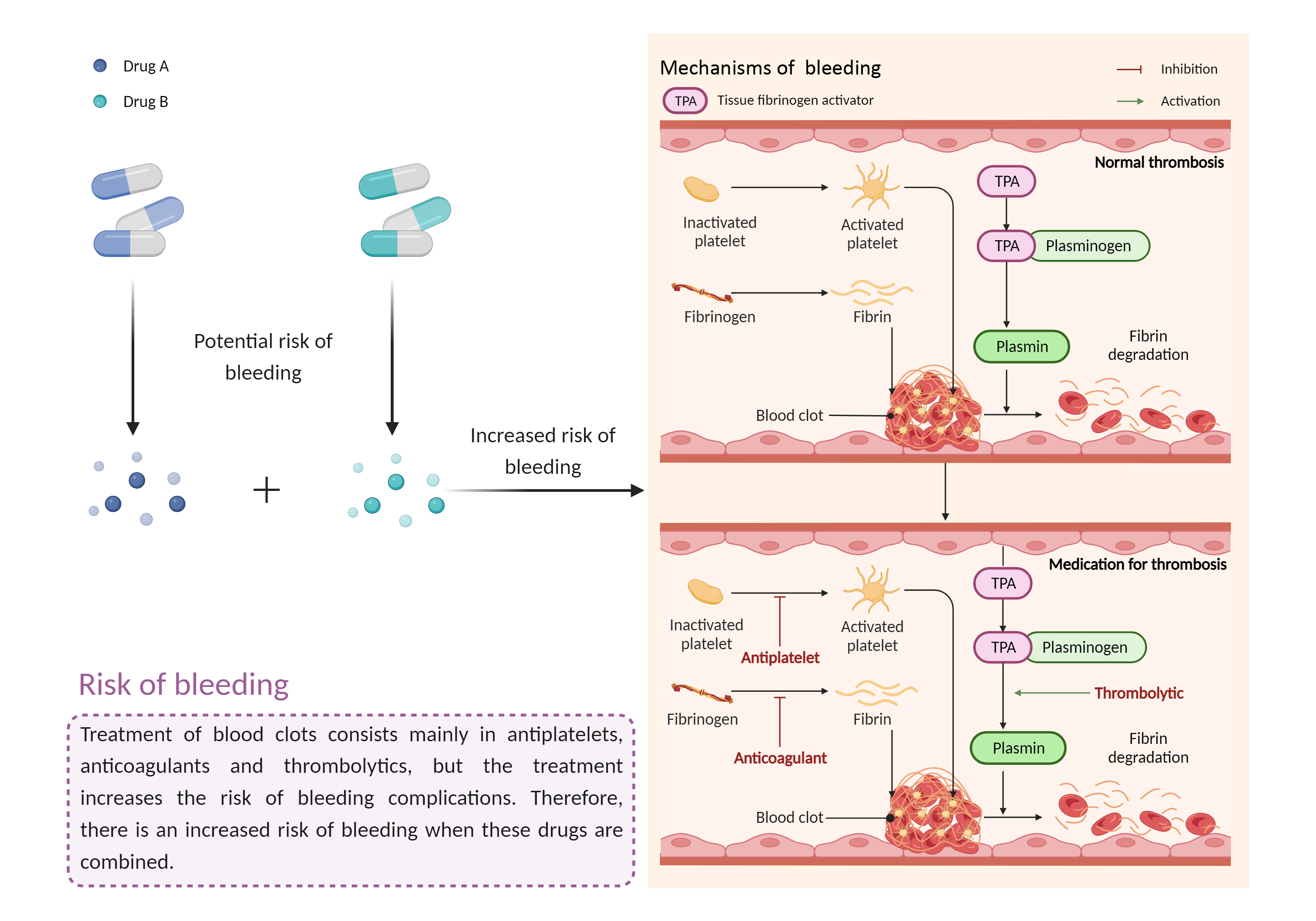 |
|||||||||
| Drug Name | Anisindione | Cefepime | |||||||
| Mechanism |
Risk of bleeding Anticoagulant |
Risk of bleeding Inhibit the growth of vitamin K-producing intestinal bacteria, inhibit the production of vitamin K-dependent coagulation factors via the methylsulfotetrazole side chain, and/or inhibit platelet activity |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Bleeding | ||||||||
| Factor Description | Patients may be at risk for bleeding when the outcome of a disease or disease treatment confounds the standard mechanisms for maintaining hemostasis. Signs or symptoms of abnormal bleeding include: bleeding that takes a long time to stop (including nosebleeds, bleeding gums, bleeding from cuts and abrasions, and menstrual bleeding); severe unexplained bruising, or bruising that becomes larger; blood in the urine or stool, etc. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Prothrombin time and INR should be monitored and the patient closely observed for signs of bleeding. The anticoagulant dose may be adjusted as indicated. Patients should be advised to notify their physicians if they experience any signs or symptoms that may indicate excessive anticoagulation, such as unusual or prolonged bleeding, bruising, coffee ground emesis, change in stool or urine color, headache, dizziness, or weakness. | ||||||||
