Details of Drug-Drug Interaction
| Drug General Information (ID: DDIURBSKL1) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Nitroprusside | Drug Info | Iodide I-123 | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Diagnostic Radiopharmaceuticals | |||||||
| Structure | |||||||||
| Mechanism of Nitroprusside-Iodide I-123 Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Interference of cell/tissue uptake Click to Show/Hide Mechanism Graph | |||||||||
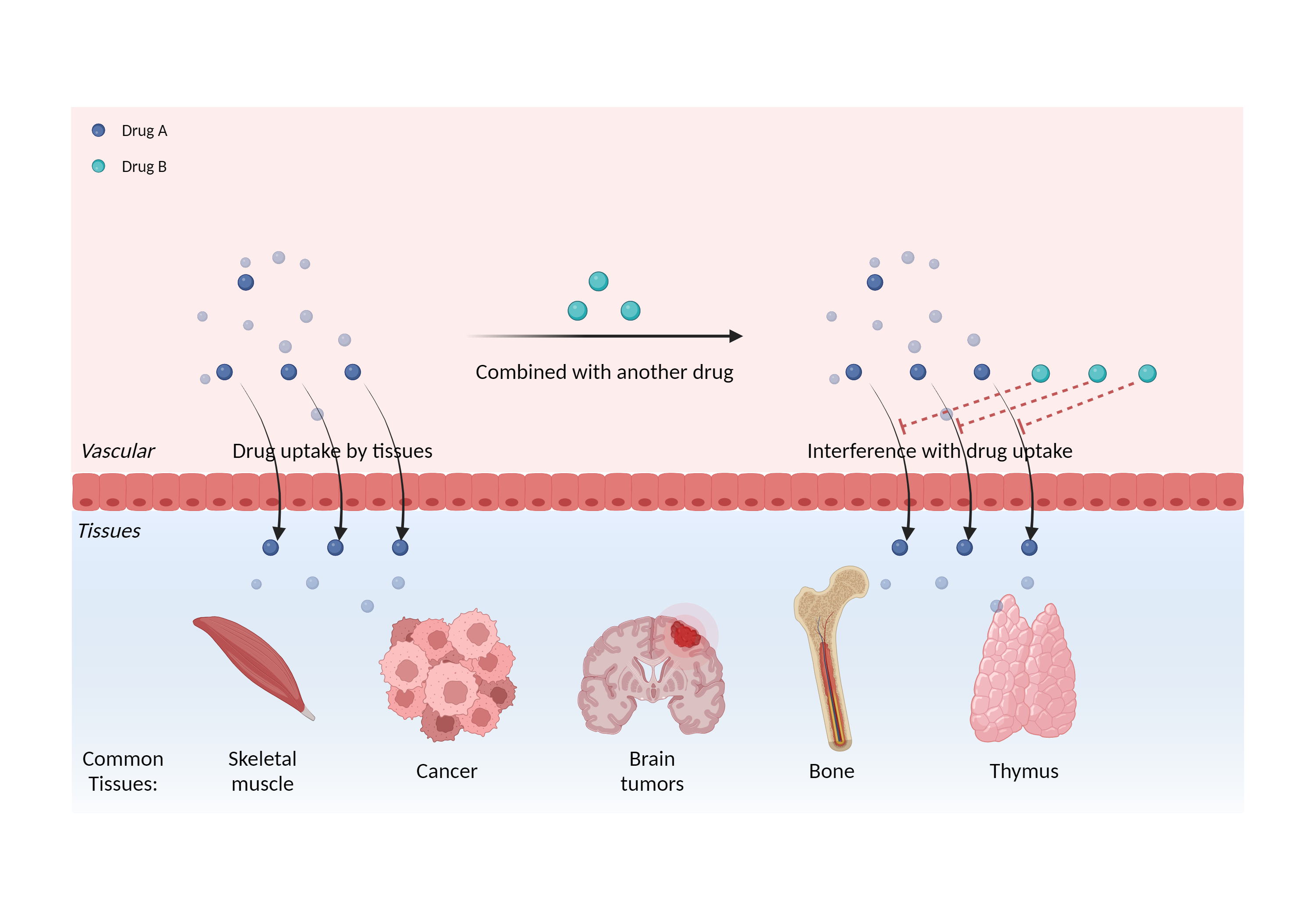 |
|||||||||
| Drug Name | Nitroprusside | Iodide I-123 | |||||||
| Mechanism | Interfere with the therapeutic effect and/or diagnostic result of radioiodides | Radioiodide | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Pharmacokinetics | ||||||||
| Factor Description | Alters the distribution of the drug, leading to changes in the plasma concentration of the drug and affecting the pharmacological effects. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Antithyroid agents such as carbimazole or propylthiouracil should generally be withheld for 1 week before administration of sodium iodide I-131 or I-123, and for a few days afterward. Salicylates, steroids, anticoagulants, antihistamines, antiparasitics, penicillins, sulfonamides, tolbutamide, thiopental, and nitroprusside should generally be withheld for 1 week. Amiodarone, lithium, and benzodiazepines should be withheld for at least 4 weeks. | ||||||||
