Details of Drug-Drug Interaction
| Drug General Information (ID: DDIUL5EQPF) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Guanabenz | Drug Info | Trichlormethiazide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Sympatholytics | Antihypertensive Agents | |||||||
| Structure | |||||||||
| Mechanism of Guanabenz-Trichlormethiazide Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
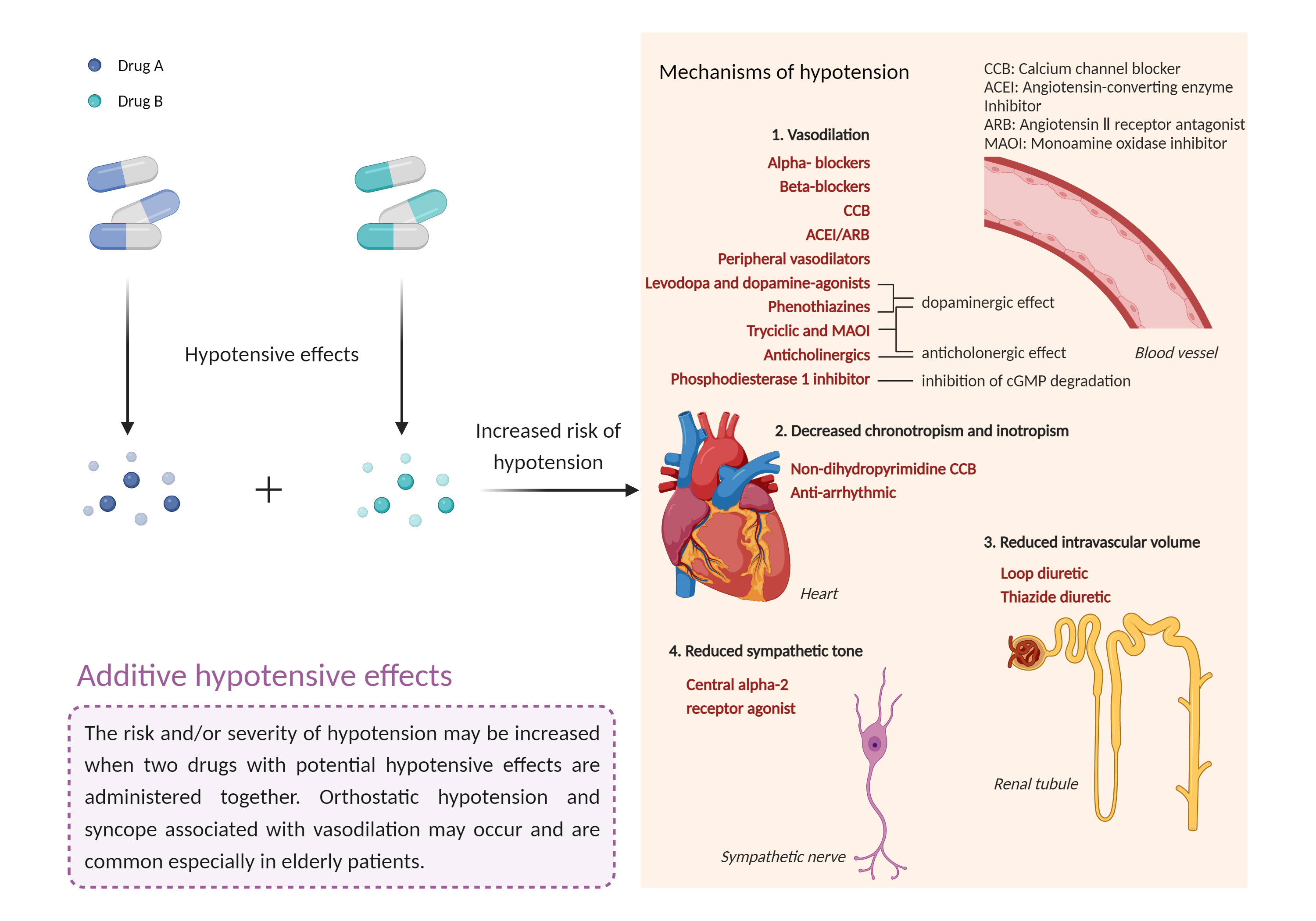 |
|||||||||
| Drug Name | Guanabenz | Trichlormethiazide | |||||||
| Mechanism |
Antihypertensive agent Alpha-2 adrenergic receptor Agonist |
Antihypertensive agent | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Adrenergic receptor alpha-2 | Structure Sequence | |||||||
| Protein Family | G-protein coupled receptor 1 family | ||||||||
| Protein Function |
Alpha-2 adrenergic receptors mediate the catecholamine-induced inhibition of adenylate cyclase through the action of G proteins. The rank order of potency for agonists of this receptor is oxymetazoline > clonidine > epinephrine > norepinephrine > phenylephrine > dopamine > p-synephrine > p-tyramine > serotonin = p-octopamine. For antagonists, the rank order is yohimbine > phentolamine = mianserine > chlorpromazine = spiperone = prazosin > propanolol > alprenolol = pindolol.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Hemodynamic responses should be monitored during coadministration, especially during the first few weeks of therapy. Patients should be advised to take the alpha-blocker at bedtime and to notify their physician if they experience dizziness or syncope while awake. | ||||||||
