Details of Drug-Drug Interaction
| Drug General Information (ID: DDIUIO759V) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Dihydroergotamine | Drug Info | Ephedrine (nasal) | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antimigraine Agents | Cns Stimulants | |||||||
| Structure | |||||||||
| Mechanism of Dihydroergotamine-Ephedrine (nasal) Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive hypertensive effects Click to Show/Hide Mechanism Graph | |||||||||
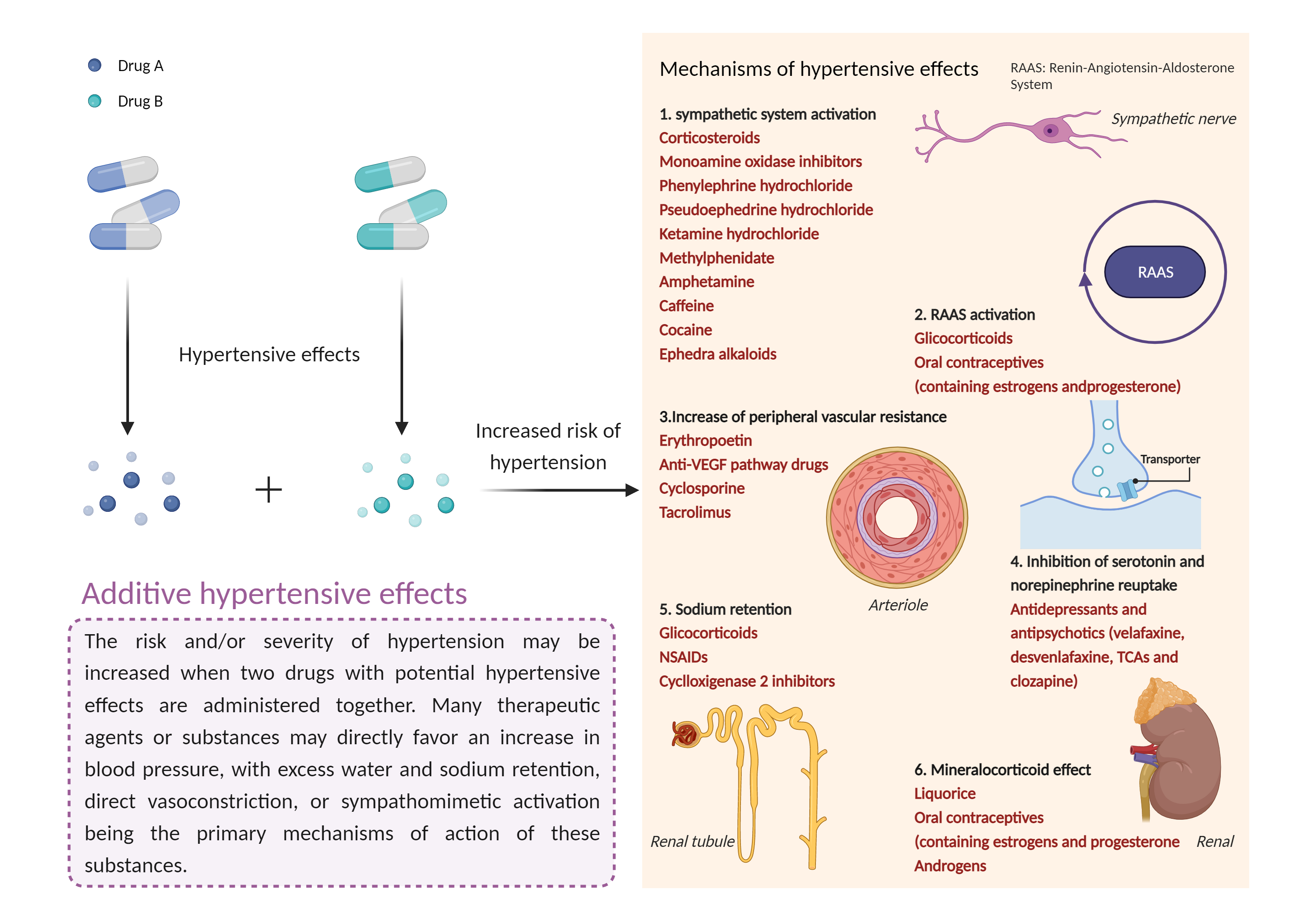 |
|||||||||
| Drug Name | Dihydroergotamine | Ephedrine (nasal) | |||||||
| Mechanism | Hypertensive effects | Hypertensive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hypertensive effects | ||||||||
| Factor Description | Hypertension is defined as a blood pressure reading above a prescribed limit (130/90 mmHg). Most people with hypertension have no signs or symptoms; others may experience: severe headache, shortness of breath, nosebleeds, severe anxiety, and feeling a tingling sensation in the neck or head. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of dihydroergotamine with other vasoconstrictive agents is considered contraindicated by the manufacturer. Patients treated with dihydroergotamine should be advised to seek immediate medical attention if they experience potential symptoms of ischemia such as coldness, pallor, cyanosis, numbness, tingling, or pain in the extremities muscle weakness severe or worsening headache visual disturbances severe abdominal pain chest pain and shortness of breath. | ||||||||
