Details of Drug-Drug Interaction
| Drug General Information (ID: DDIUC9M6WY) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Salsalate | Drug Info | Teriflunomide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiinflammatory Agents | Selective Immunosuppressants | |||||||
| Structure | |||||||||
| Mechanism of Salsalate-Teriflunomide Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
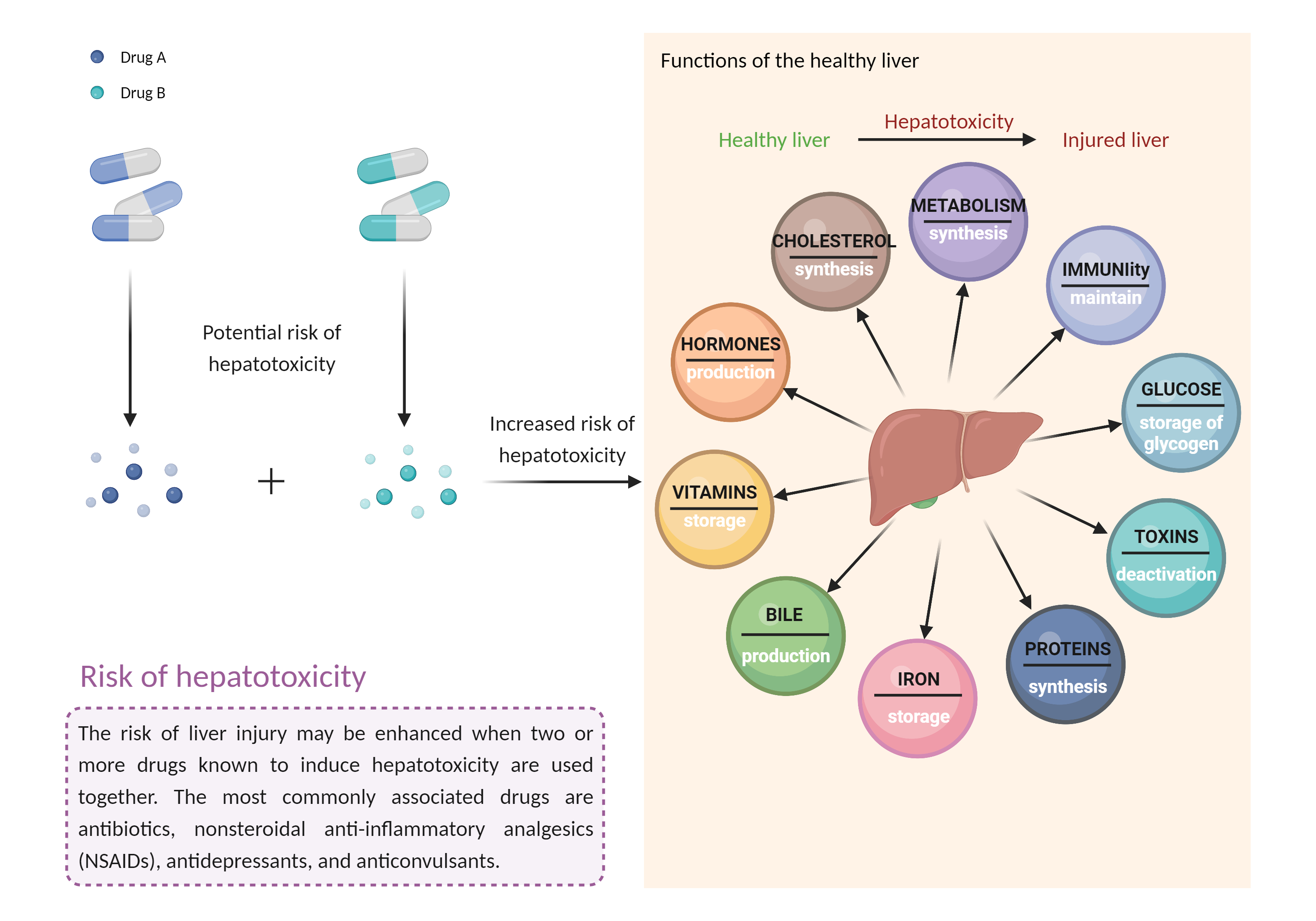
| Increased risk of hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Salsalate | Teriflunomide | |||||||
| Mechanism | Hepatotoxicity | Hepatotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Patients receiving leflunomide or teriflunomide in combination with NSAIDs should be closely monitored for hepatotoxicity. Liver enzymes and bilirubin should be measured prior to initiation of leflunomide/teriflunomide therapy and at least monthly for the first six months of treatment and every 6 to 8 weeks thereafter. Patients with preexisting liver disease or elevated baseline liver enzymes (i.e., ALT greater than two times the upper limit of normal) should not receive leflunomide or teriflunomide. Patients who develop elevated serum ALT greater than three times ULN while receiving these medications should discontinue treatment and be given washout procedures with cholestyramine or activated charcoal to accelerate elimination of leflunomide's active metabolite from plasma, which otherwise may take up to two years. | ||||||||
