Details of Drug-Drug Interaction
| Drug General Information (ID: DDITM5JFB1) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Simeprevir | Drug Info | Enasidenib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiinfective Agents | Antineoplastics | |||||||
| Structure | |||||||||
| Mechanism of Simeprevir-Enasidenib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
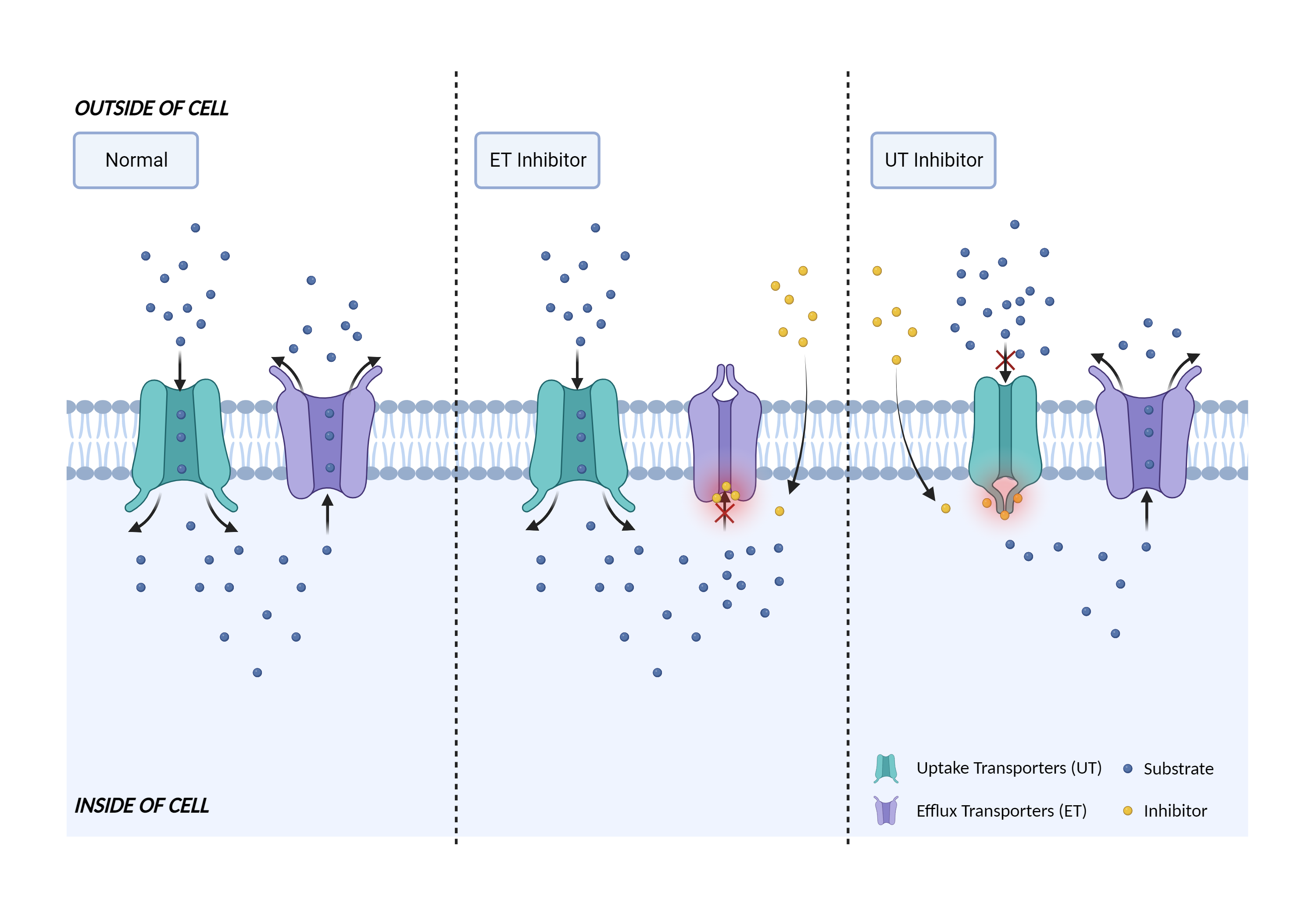
| Transporter inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Simeprevir | Enasidenib | |||||||
| Mechanism | OATP1B1 substrate | OATP1B1 inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Liver organic anion transporter 1 |
×
Structure
Sequence
MDQNQHLNKTAEAQPSENKKTRYCNGLKMFLAALSLSFIAKTLGAIIMKSSIIHIERRFEISSSLVGFIDGSFEIGNLLVIVFVSYFGSKLHRPKLIGIGCFIMGIGGVLTALPHFFMGYYRYSKETNINSSENSTSTLSTCLINQILSLNRASPEIVGKGCLKESGSYMWIYVFMGNMLRGIGETPIVPLGLSYIDDFAKEGHSSLYLGILNAIAMIGPIIGFTLGSLFSKMYVDIGYVDLSTIRITPTDSRWVGAWWLNFLVSGLFSIISSIPFFFLPQTPNKPQKERKASLSLHVLETNDEKDQTANLTNQGKNITKNVTGFFQSFKSILTNPLYVMFVLLTLLQVSSYIGAFTYVFKYVEQQYGQPSSKANILLGVITIPIFASGMFLGGYIIKKFKLNTVGIAKFSCFTAVMSLSFYLLYFFILCENKSVAGLTMTYDGNNPVTSHRDVPLSYCNSDCNCDESQWEPVCGNNGITYISPCLAGCKSSSGNKKPIVFYNCSCLEVTGLQNRNYSAHLGECPRDDACTRKFYFFVAIQVLNLFFSALGGTSHVMLIVKIVQPELKSLALGFHSMVIRALGGILAPIYFGALIDTTCIKWSTNNCGTRGSCRTYNSTSFSRVYLGLSSMLRVSSLVLYIILIYAMKKKYQEKDINASENGSVMDEANLESLNKNKHFVPSAGADSETHC
|
|||||||
| Gene Name | OATP1B1 | ||||||||
| Uniprot ID | SO1B1_HUMAN | ||||||||
| KEGG Pathway | hsa:10599 | ||||||||
| Protein Family | Organo anion transporter (TC 2.A.60) family | ||||||||
| Protein Function |
Mediates the Na(+)-independent uptake of organic anions such as pravastatin, taurocholate, methotrexate, dehydroepiandrosterone sulfate, 17-beta-glucuronosyl estradiol, estrone sulfate, prostaglandin E2, thromboxane B2, leukotriene C3, leukotriene E4, thyroxine and triiodothyronine. Involved in the clearance of bile acids and organic anions from the liver.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised when enasidenib is used concurrently with medications that are substrates of OATP 1B1/1B3 and/or BCRP, particularly those with a narrow therapeutic range. Dosage adjustments as well as clinical and laboratory monitoring may be appropriate for some drugs whenever enasidenib is added to or withdrawn from therapy. | ||||||||
